Course Authors
Elizabeth M. Ross, M.D., L.D.N.
Release Date: 11/11/2002
Upon completion of this Cyberounds®, you should be able to:
Identify patients at risk for or with iron deficiency
Discuss the causes of increased iron requirements
Select treatment for iron deficiency and be able to identify high-iron foods.
Iron deficiency is a fairly prevalent problem in populations typically seen by primary care providers, especially among menstruating women. Of women aged 20 to 49, 5% have iron deficiency anemia and 11% have iron deficiency without anemia.(1) Aside from its role in oxygen transport and delivery, iron serves as a cofactor for many physiologically important enzymes including those involved in oxidative metabolism, dopamine and DNA synthesis, and free radical formation in neutrophils. For this reason, in addition to causing anemia, iron deficiency may affect work capacity, neurotransmitter function, and immunologic and inflammatory defenses.
Iron Deficiency Risk
Risk for iron deficiency is a function of levels of iron loss, iron intake, iron absorption and physiologic demands. Women in their childbearing years have greater iron needs than men as a result of menstrual blood losses, iron demands of the developing fetus and blood loss during childbirth. In addition, uterine fibroids may cause heavy and prolonged menses, leading to increased blood loss. Iron deficiency may also be caused by other sources of blood loss including gastrointestinal bleeding from gastritis and peptic ulcer disease, neoplasms, inflammatory bowel disease, parasitic infections (more common in third world populations than developed countries) and hemorrhoids. Chronic blood loss may occur from the urinary tract or pulmonary systems as well.
In order to avoid the development of iron deficiency, dietary iron intakes must keep pace with demands. Recommended dietary allowances for men over the age of 19 and women over the age of 51 are 8 mg per day; those for women ages 19 to 50 are 18 mg per day.(2) In American diets, major contributors to iron intakes are meat, poultry, fish, nuts and seeds, legumes and bean products, green leafy vegetables, raisins, whole grains and fortified cereals. Iron contents of some commonly consumed high iron foods are shown in Table 1.
Table 1. Iron Content of Selected High-iron Foods.
| Food | Portion Size | Iron (mg) |
| Total® cereal | 1 cup | 18 |
| Grape Nuts® cereal | 1/2 cup | 8.2 |
| Instant plain oatmeal | 1 packet | 6.7 |
| Wheat germ | 1 ounce (1/4 cup) | 2.6 |
| Broccoli | 1 medium stalk | 2.1 |
| Baked potato | 1 medium | 2.7 |
| Spinach | 1 cup raw | 0.8 |
| Dried peach | 5 halves | 2.6 |
| Raw tofu | 1/2 cup | 4 |
| Lentils | 1/2 cup | 3.3 |
| Kidney beans | 1/2 cup | 2.6 |
| Chickpeas | 1/2 cup | 2.4 |
| Beef chuck | 3 ounces | 3.2 |
| Dark meat turkey | 3 ounces | 2.0 |
| Blackstrap molasses | 1 tablespoon | 5.0 |
Physiology of Iron Absorption
Iron absorption is not directly correlated to iron intake. As physiologic iron levels decrease, gastrointestinal absorption of iron increases. The bioavailability of iron, or the percentage of dietary iron absorbed and ultimately physiologically available, also varies, depending on the dietary source of the iron and other foods consumed at the same time as the iron-containing foods.
Heme and non-heme iron are absorbed by different receptors on the intestinal mucosa. Iron bound to heme is highly absorbable -- 40% of iron from animal sources is in this form. The absorption of non-heme iron can be increased or decreased by various factors. Phytates, or inositol phosphate salts which store minerals in plant matter, bind iron in the lumen of the intestine and decrease its absorption. Polyphenols in tea, coffee, cocoa, spinach and oregano inhibit iron absorption as well. Iron is best absorbed in its ferrous form, and thus reducing substances, such as ascorbic acid in fruits, vegetables and fortified cereals, increase iron absorption. Calcium inhibits the absorption of both heme and non-heme iron by an unknown mechanism and, interestingly enough, epidemiologic studies show a correlation between intake of milk and prevalence of iron deficiency.
Identifying Patients at Risk for or with Iron Deficiency
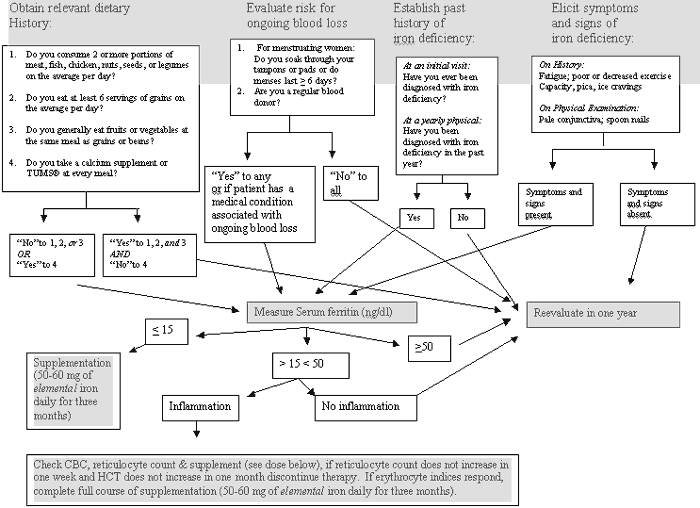
During a yearly physical or health maintenance visit, four pieces of information concerning iron may be ascertained to gage risk for iron deficiency:
- Relevant dietary history
- Excessive menstrual loss or other cause of increased iron requirements
- Previous history of iron deficiency
4. Current signs or symptoms of iron deficiency.
Relevant Dietary History
The goal of asking about iron intake in a medical setting should not be to ascertain the exact iron content of a patient's diet in milligrams per day but rather to flag patients who might be at risk for iron deficiency based on their usual intakes or their usual intakes of other nutrients that may affect iron absorption. The following four questions should accomplish this objective:
- Do you consume two or more portions of meat, fish, chicken, nuts, seeds or legumes per day?
- Do you eat at least six servings of grains per day?
- Do you generally eat at least one serving of fruits or vegetables containing foods at the same meal as grains or beans?
- Do you take a calcium supplement or OTC antacid at every meal?
The first two questions ask about consumption of iron containing foods. The third and fourth questions ask about consumption of foods that may affect iron absorption. Patients answering no to one of the first three questions or yes to the last question may be at risk for iron deficiency and a laboratory evaluation should be completed in these patients. Other patients should be reevaluated periodically.
Excessive Menstrual Losses or Other Causes of Increased Iron Requirements
Menstrual losses can be roughly quantified by asking the following questions:
- Do you soak through your tampons or pads?
- Do your menses last longer than six days?
Patients answering yes to either of these questions may be at risk for iron deficiency and laboratory evaluation should be completed in these patients. Other patients should be reevaluated in a year. This high frequency of reevaluation is recommended in perimenopausal women who might be experiencing large changes in the quantity of blood loss during menses.
Other causes of increased iron requirements include pathologic gastrointestinal, urinary and pulmonary losses. Any patient with one of these sources of blood loss should receive further evaluation and treatment. A final non-pathologic cause of increased iron requirements is frequent blood donation. Any patient who donates blood regularly should be screened for iron deficiency during a yearly physical examination.
Previous Diagnoses of Iron Deficiency
Though treatment for iron deficiency is generally completed over a relatively short period of time with a short course of iron supplementation, the cause of the deficiency may be ongoing and deficiency may recur. For this reason, patients who have been diagnosed with iron deficiency in the past should be reevaluated for iron deficiency periodically. Patients should be asked about a history of iron deficiency at new patient visits and yearly physicals, and those with deficiency should receive a laboratory evaluation.
Signs and Symptoms of Iron Deficiency
During the review of systems, symptoms of iron deficiency should be elicited; signs of iron deficiency should be looked for during the physical examination. Symptoms of iron deficiency include fatigue, poor exercise tolerance, pica (compulsive eating of non-food items) and pagophagia (compulsive eating of ice); signs of iron deficiency include pale conjunctiva and spoon nails. Patients having any of these signs and symptoms should receive a laboratory evaluation for iron deficiency.
Laboratory Evaluation of Iron Deficiency
Measurement of serum ferritin is recommended for diagnosis of iron deficiency. Serum ferritin concentrations reflect body iron stores (one microgram per liter of serum ferritin concentration is equivalent to approximately 10 mg of stored iron). As compared to the gold standard of a bone marrow biopsy, serum ferritin is a sensitive and specific indicator of iron depletion. Levels below 15 µg/L are 75% sensitive and 98% specific for iron deficiency.(3) Because serum ferritin is, however, an acute phase reactant, chronic infection, inflammation or diseases causing tissue and organ damage can raise its concentration independent of iron status, thus masking depleted tissue stores.
Transferrin saturation (serum iron concentration divided by the total-iron binding capacity [TIBC] x 100) reflects the extent to which iron binding sites are vacant on transferrin and is another commonly used measure of iron deficiency. Overall, the measure does not perform as well as ferritin: among non-pregnant women of childbearing age, the sensitivity of low transferrin saturation (16%) for iron deficiency (as defined by no stainable bone marrow iron) is only 20% and the specificity is 93%. Like ferritin, factors other than iron status can affect results of this test. Serum iron varies diurnally (higher in the AM, lower in the PM), increases after meals and is decreased by infection and inflammation. Inflammation, chronic infection, malignancies, liver disease, nephrotic syndrome and malnutrition can reduce TIBC, while oral contraceptive use and pregnancy can increase it.
Given the high specificity of a low ferritin for iron deficiency, in an otherwise healthy patient, it is not necessary to check a CBC (complete blood count) as part of an iron deficiency evaluation since the results of this test will not alter management. If ferritin is low, iron deficiency should be treated regardless of whether or not anemia is present because, with iron stores depleted, symptoms of deficiency may be present before the development of an overt anemia. In the presence of an inflammatory or infectious state, however, when iron risk factors for or symptoms of iron deficiency are present but ferritin is in the normal range, a CBC and a reticulocyte count may be quite helpful. If iron deficiency is indeed present, erythrocyte indices should improve with iron administration and a therapeutic trial of supplementation will help to confirm or rule out iron deficiency.
Treatment of Iron Deficiency
A ferritin of <15 µg/L is indicative of iron deficiency and patients with levels in this range should receive a course of replacement therapy. Iron is best absorbed in its ferrous form and ferrous salts of iron are generally used for oral supplementation. Ferrous sulfate, succinate, lactate, fumarate, glycine sulfate, glutamate and gluconate are all about equally well absorbed. Standard doses are 50 mg to 60 mg of oral elemental iron twice daily. Vitamin C taken concurrently with the iron will increase absorption. In the event of gastrointestinal distress, the dose should be reduced by ½ and oral supplementation continued.
Enteric coated or delayed-release preparations should not be used -- with these preparations iron is released distally in the small intestine or in the colon where it is not well absorbed. Intravenous administration is sometimes necessary in cases of intolerance of oral supplements. Iron dextran has been associated with rare anaphylactic reactions, but newer formulations have a lower incidence of side effects and may be given in the office without undue risk of complications.(4)
The Centers for Disease Control and Prevention (CDC) recommends a three-month course of therapy for the treatment of iron deficiency. In the presence of anemia, reticulocyte counts will begin to rise after a few days of supplementation and will peak in approximately seven days. Hemoglobin will begin to rise after 10 to14 days and will generally normalize in two months.
Some authors recommend continuing supplementation for six to twelve months; however, as iron status improves, a lower proportion of the supplement dose is absorbed and the benefits of supplementation are thus reduced as the course of therapy is lengthened. During the course of supplementation, patients should be advised concerning higher iron diets and any correctable cause of blood loss should be addressed. Those experiencing ongoing uncorrectable blood loss may benefit from daily low dose supplementation after replacement regimens are completed and from incorporating more high iron foods into their regular diets.(5)