Low Carb v. High Protein Diets: 2019 Update
Course Authors
Edward Saltzman, M.D., is Academic Dean for Education at the Friedman School of Nutrition Science and Policy, Associate Professor at the School of Medicine, and scientist at the Energy Metabolism Laboratory in the Jean Mayer USDA Human Nutrition Research Center on Aging, Tufts University, Boston, MA.
Release Date: 09/03/2019
Learning Objectives
Upon completion of this Cyberounds®, you should be able to:
Describe the mechanisms by which macronutrient composition could influence weight loss;
Discuss the potential risks associated with low carbohydrate diets;
Identify classes of patients in whom low carbohydrate/high fat or high carbohydrate diets could worsen cardiovascular risk factors.
Approximately two-thirds of Americans are now classified as overweight or obese.(1) As obesity prevalence has increased throughout the society, clinical understanding of the nature of obesity has evolved: what was once thought of throughout the 20th century as a moral failing of overeating has since changed into a singular view of hormonal dysregulation (in particular defects in insulin sensitivity). Despite this paradigm shift from moral to physiologic understanding, outcomes have continued to worsen, suggesting that a single mechanism is not the cause.(1) Several genetic and anthropological researchers suggest that genetic predisposition to a hunter-gatherer or farming based metabolism may play a role in these changes, as diets have evolved alongside our economies and ways of life.
In a reasonable attempt to combat these changes, both practitioners and patients alike have turned to different diets: 44% of women and 29% of men reported trying to lose weight at any given time.(2) While losing weight is difficult, maintaining weight loss is even more challenging. Approximately 85% of those who lose weight will regain their original weight within five years. It comes as no surprise that a wide variety of weight loss diets have appeared in the media. Many of these approaches entail a significant change in the proportion of dietary macronutrients (carbohydrate, protein and fat) in comparison to the "average" U.S. diet. Among the most popular current recommendations are programs that limit dietary carbohydrates, also known as "low carb" diets.
Goal: Energy Intake = Energy Output
Changes in body weight require an imbalance between energy intake from food and energy expenditure. Energy expenditure can be divided into three components: resting energy expenditure (REE), which accounts for approximately two-thirds of daily expenditure and is largely determined by the amount of lean body mass; the thermic effect of feeding (TEF), approximately 10-15% of daily expenditure resulting from the energy stimulated by the intake, digestion and absorption of nutrients; and energy spent on physical activity (Figure 1).

Figure 1. Components of Energy Expenditure.


Based on current reviews of controlled feeding studies of both obese and lean subjects (important in the discussion of weight loss given U.S. obesity rates), people with obesity may expend comparable energy for physical activity despite overall less physical activity because expenditure is proportional to body weight. In addition, resting energy expenditure is greater in obese subjects due to more metabolically active fat-free mass in addition to greater body fat.(3)
Resting energy expenditure is greater in obese subjects.
Energy intake is influenced by numerous factors, including diet, hunger and emotions, as well as environmental factors such as food availability and social norms. Many dietary factors also influence food intake (Table 1).

Table 1. Dietary Factors Proposed to Influence Energy Intake.
|

Dietary macronutrient composition is expressed either as an absolute amount of each macronutrient, or as a percent of the contribution of each macronutrient to the total energy content of the diet. When expressed as a percent of dietary energy, it is important to remember that any reduction in carbohydrate will necessarily increase the percentage of fat and/or protein in the diet. To assess the effects of a low carbohydrate diet, therefore, the role of the other increased macronutrients must also be considered.
Macronutrient Effects on Energy Expenditure and Weight Loss
Food energy must ultimately be stored as body energy (also known as nutrient partitioning into fat, glycogen or protein), used in the conversion to chemical energy (e.g., ATP), released as heat or excreted (as in glycosuria with uncontrolled diabetes). It would be advantageous if a particular diet composition could influence its own oxidation, which we could then measure as an increase in energy expenditure. In the most ideal weight loss program, the diet composition itself would selectively cause loss of body fat but not of other tissues.
In studies where subjects were fed weight-maintaining diets, little effect of macronutrient composition on the total energy needed to maintain weight was observed.(4) Where research subjects were deliberately overfed beyond their maintenance needs, the major factor in determining deposition of body fat was the amount of excess energy beyond maintenance needs. The macronutrient composition had a relatively minor impact on the storage of excess energy as fat.(5)
When individual macronutrients are compared, there are indeed some differences in the efficiency of storing excess dietary energy as body fat (an undesirable effect for weight loss): protein is stored less efficiently than carbohydrate, which in turn is stored less efficiently than fat. These differences are narrow, however: about 90-95% of ingested fat is stored compared to 80-85% of carbohydrate.(5) Since most diets are composed of mixtures of macronutrients, these differences in efficiency are unlikely to result in important changes in expended energy. One explanation for this difference in energy storage is that the macronutrients have different TEFs: while only a small percentage of fat energy is "burned off" after ingestion, the TEF of protein can be more than 25% of ingested calories (carbohydrate "burn off" is intermediate). These differences in TEF could result in marked variability in body weight over the long-term only in extreme dietary compositions of macronutrients. The typical Western diet contains enough mixture of macronutrients that this effect would be negligible.
Macronutrient composition had relatively minor impact on storage of excess energy as fat.
In a review of trials prior to 2002, Eisenstein et al. evaluated the influence of hypocaloric diets with increased dietary protein (mostly associated with decreased carbohydrate) on loss of body weight and loss of lean mass.(6) In trials where identical calories were provided, no consistent benefit of increased protein on weight loss was found and these higher protein diets did not spare lean mass during weight loss. Further, diets that induced ketosis had no consistent effect on preservation of lean mass. More recent studies, however, have contradicted these findings and shown that increased proportions of dietary protein have increased fat-free body mass in periods of both weight loss and weight gain.(7)(8)(9) Additionally, Wycherley et al. demonstrated through a meta-analysis investigating variation in protein ingestion in low-fat diets that increases in dietary protein can increase REE (although the absolute value is again negligible in the context of the typical diet).(10)
Given the current interest in popular culture on ketogenic and Atkins-related diets, particular focus on the effects of dietary fat and carbohydrate composition on weight loss is warranted. Previous work by Bravata et al. reviewing low carbohydrate diets revealed no advantage with respect to weight loss or preferential fat loss, although not all studies compared isocaloric diets.(11) According to the researchers, the most important factors contributing to weight loss were the degree of energy restriction and the duration of the study. In addition, those with greater body mass index lost more weight.(3)
Recent work by Hall and Guo offer an updated analysis of low carbohydrate diets. In reviewing these claims of the "carbohydrate-insulin model" (whereby insulin release during carbohydrate ingestion preferentially shunts glucose towards fat storage, as opposed to higher-percentage fat ingestion promoting release of free fatty acids), the authors conducted an exhaustive review and meta-analysis of diets varying in fat and carbohydrate composition but equal in protein. These studies included only controlled feeding trials where food was provided to subjects. In effect, the "carbohydrate-insulin model" was not supported; lower-fat diets produced greater energy expenditure but the overall magnitude of differences was clinically insignificant. Total body fat composition was also found to slightly favor low-fat diets.(3)
Some advocates of low carbohydrate diets have claimed that fat loss occurs with these diets while sparing lean mass. Layman and colleagues provided two reduced isocaloric diets differing in protein (mean 125 vs. 68 g/day) and carbohydrate (mean 171 vs. 246 g/day) to overweight women for 10 weeks.(12) They found no difference in the total mean weight lost (7.53 vs. 6.95 kg in higher vs. lower protein, respectively). Body composition assessed by dual x-ray absorptiometry revealed that there were no statistically significant differences in the amount of fat or lean mass lost between the diets, although there was a trend toward a lower loss of lean mass for those on the higher protein diet (loss of lean mass 0.88 vs. 1.21 kg for the higher vs. lower protein diets, respectively). Only when lost weight was expressed as a ratio of fat:lean mass did the difference become statistically significant.
The amount of weight lost by subjects in the study by Layman is very typical of that experienced among free-living persons attempting to lose weight, although it is possible that subjects may have needed to lose considerably more weight to allow the differences in nutrient partitioning to become statistically significant. The relatively small differences in loss of fat and lean mass observed between the two diets suggest that we need to remain cautious before extrapolating from statistical significance to meaningful clinical significance.
Macronutrient Effects on Energy Intake and Weight Loss
Thus far it appears that no clear advantage is conferred by a particular macronutrient composition on energy expenditure or nutrient partitioning. Previously cited studies mandate isocaloric dietary regimens so that differences in macronutrient content can be isolated without confounding effects from differences in total energy intake; however, it is possible that a particular macronutrient composition might result in lower spontaneous energy intake, reduced hunger or greater satiety. Since hunger is a frequent complaint of dieters, it is possible that macronutrient manipulation might result in increased ability to tolerate a calorie deficit.
Multiple studies have focused on the effect of lower carbohydrate intake on overall decreased energy consumption. Foster et al. educated moderately overweight subjects to follow either an ad libitum low carbohydrate diet (subjects tried to reduce carbohydrates to an initial goal of 20 g/day and were given a copy of Dr. Atkins New Diet Revolution) vs. a low-fat diet with specified calorie restriction of 1200-1500 calories/day.(13) The low carbohydrate diet resulted in significantly greater weight loss at six months; however, at 12 months (end of the study), the weight difference between lower and higher carbohydrate diet groups was no longer significant. Findings from Samaha et al. support this evidence that (at least in the short-term) low carbohydrate diets result in greater weight loss.(14) In this case, extremely obese subjects (mean BMI 43 kg/m2) were selected for either reduced-carbohydrate (30 g/day) or energy-restricted (500 calories/day below expected maintenance needs, <30% calories from fat) diets: after six months, the low carbohydrate diet resulted in a significantly greater weight loss, 5.8 kg in comparison to 1.9 kg on the more conventional diet.
Total body fat composition found to slightly favor low-fat diets.
In addition to the effects of reducing carbohydrate intake, changes in dietary protein have been shown to affect energy intake. Skov et al. utilized a novel method to control macronutrient content while allowing free living subjects to select food (better simulating a natural consumer environment that strictly controlling all intake).(15) Over a 6-month period, subjects were able to choose food from a small shop created by the investigators. The selected food, as well as total amount consumed, was also documented by the investigators. After six months, subjects eating a diet with 25% protein/45% carbohydrate/30% fat consumed fewer calories and lost more weight (8.9 kg) compared to those consuming a diet with 12% protein and a commensurate increase in carbohydrate (loss of 5.1 kg). In this study, the increased protein content and lower carbohydrate content, while still in the range consumed by many Americans, resulted in decreased food intake and superior weight loss.
The mechanisms by which low carbohydrate diets favorably reduce energy intake and body weight remain speculative. One potential mechanism relies on the concept of the glycemic index, which describes the blood glucose response to food in comparison to known carbohydrate loads (standards such as dextrose or white bread). In short-term studies, lower glycemic index meals suppressed appetite and reduced intake later in the same day in comparison to higher glycemic index meals.(16) Proponents suggest that the insulin response following glucose absorption mediates subsequent food intake; the blunted insulin response to a lower glycemic index meal translates into decreased food intake. While short trials of this mechanism appear promising, longer-term data have not yet confirmed this effect on weight loss. In the Samaha and Foster studies, as well as in others, improvements in fasting glucose, insulin or insulin sensitivity are not consistently observed in response to low carbohydrate diets.(11)(13)(14)(17)
Differences in satiation between macronutrients have been cited in prior research: Eisenstein et al. showed that protein is superior to carbohydrate, which is in turn superior to fat in feeling full after a single meal.(6) If this effect persists over weeks to months, it is possible that increased protein content might allow a greater overall decrease in intake. Indeed, Ebbeling et al. further demonstrated in a randomized control trial that obese subjects consuming controlled amounts of carbohydrates experienced hormonal changes that lend themselves to greater satiation.(18) Ghrelin, a hormone produced in part by the stomach which leads to increased hunger, increased fat deposition and lower energy expenditure, was found in lower concentrations in subjects consuming lower carbohydrate diets. Additionally, leptin (an adipocyte-generated hormone that acts to inhibit hunger) was also found to decline in low-carbohydrate participants, suggesting greater sensitivity to this hormone. Authors of this study, while also demonstrating greater weight loss with lower carbohydrate consumption in line with the "carbohydrate-insulin model", speculate that based on additional prospective studies, greater declines in leptin offer the highest likelihood of preventing weight regain. (18)
Ketosis has been reported to suppress appetite, and this remains a potential mechanism that might decrease intake. While reports of this effect are largely anecdotal, Paoli et al. have proposed a more unified theory that accounts for both the anorexigenic (appetite-reducing) and orexigenic (appetite-stimulating) signals. They postulate that ketogenic diets, through increased circulating ketone levels (generally beta hydroxybutarate), increase adiponectin and GABA levels in the brain, leading to decreased reactive oxygen species in circulation and overall increased hunger; these effects, however, are overcome by the anorexigenic increase in circulating post-meal free fatty acids (thus reducing cerebral neuropeptide Y [NPY]) and decreasing circulating ghrelin.(19)
Sumithran et al. have demonstrated this complex interaction of hormonal regulators of hunger and appetite as well as the subjective experiences of patients. In a 10-week trial of ketogenic diets in overweight and obese men (during which participants maintained ketosis for eight weeks with a return to non-ketosis for the remaining two weeks), subjective hunger was lower during the ketotic phase than during refeeding. During this period of statistically significant weight loss, ketotic subjects were found to have ghrelin suppression After re-feeding, however, these levels rose significantly.(20) Additional markers of metabolism, including mean fasting and postprandial glucose, followed a similar pattern.
Ketosis reported to suppress appetite.
Small-scale trials such as that performed by Johnstone et al. reported the effect of decreased overall energy intake in ketogenic participants: of 17 obese men enrolled in a residential trial where foods were controlled, those assigned to a "ketogenic diet" (4% carbohydrates) demonstrated lower ad libitum energy intake than their higher-carbohydrate peers. Additionally, the ketogenic participants had significant reductions in subjective hunger as well as objective weight loss. While only performed in the short-term (4-week period), the results show a macronutrient-derived approach to reduction in ad libitum energy consumption.(22) Additional studies are still needed to examine these effects, as well as the clinical implications on weight loss of ketogenic diets.
Additionally, because the range of foods that can be consumed while following a low carbohydrate diet is limited, it is possible that the reduced number of food choices results in better diet compliance. This phenomenon, termed stimulus narrowing, is essentially the elimination of typically over-consumed foods. This principle is best illustrated by very low calorie formula diets where no other food is consumed. In a practical sense, patients eliminate the mental stress of dieting by removing choice from the act of eating.
Macronutrients and End Organ Changes
It appears that reducing carbohydrate to very low levels, as with the Foster and Samaha studies, may confer an advantage over several months. If hunger is suppressed, and intake is reduced, this macronutrient mix might be an effective strategy for some. Critics of this strategy, however, cite potential effects on bone health, cardiovascular risk factors and other end organ changes from such high fat, high protein diets.
High protein intake, especially of sulfur-containing amino acids, results in increased bone resorption and promotes calciuria.(21) A potential mechanism of this change is the role of bone in buffering acid loads induced by amino acids.(21) However, a buffering effect of other dietary constituents such as fruit and vegetables has also been observed, and adequate calcium intake may also minimize deleterious effects on bone.(23)(24) At present, the effects of high protein diets on bone health remain unresolved.
Cuenca-SA!nchez et al. provided a detailed review of the effects of high protein diets on not only bone health, but also kidney function.(25) Several studies have demonstrated the effect of worsening GFR with high protein diets on patients with pre-existing renal dysfunction, which may tend to occur in patients suffering from obesity (as well as the elderly, who naturally have lower GFRs). High protein diets, however, do not seem to have the same effect on otherwise healthy individuals. The authors make an important point that with high protein intake there is a natural increase in solute excretion (urea, wastes, etc.), requiring increased total water intake to prevent dehydration. Additionally, high protein diets have been seen to have a positive correlation with kidney stone formation, especially in those predisposed to nephrolithiasis.(25)
As per the review by Kosinski and Joynavaz,(26) ketogenic diet may be associated with some improvements in cardiovascular risk factors, such as obesity, type 2 diabetes and HDL cholesterol levels, but these effects are usually limited in time. Also, there was inconclusive effect on insulin resistance and sensitivity, as some studies found that Insulin resistance could be a potential negative effect, but others have shown improvements in insulin sensitivity with ketogenic diets.
In general, the low carbohydrate weight loss diets described above, as well as others, have not resulted in increased total and LDL cholesterol predicted by some (Figure 2).
Low carbohydrate weight loss diets have not resulted in increased total and LDL cholesterol.

Figure 2. Effects of Low Carbohydrate and Low-Fat Diets on Blood Lipids.
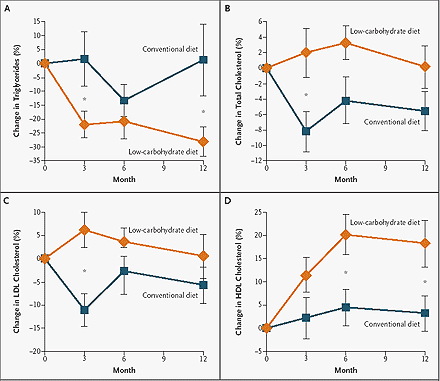
From Foster GD, Wyatt HR, Hill JO, et al. A randomized trial of a low-carbohydrate diet for obesity. N Engl J Med 2003;348:2082-90. Copyright 2003 Massachusetts Medical Society. All rights reserved.

With regard to HDL cholesterol, greater increases (vs. smaller increases or decreases) have been observed, although HDL subfractions remain unclear in some studies.(27) The increase in triglycerides, at times observed with diets high in carbohydrates, is also not seen with reduced carbohydrate diets. This does not, however, mean that low carbohydrate diets will not adversely influence blood lipids: as noted in an editorial accompanying the Foster and Samaha studies, consumption of high levels of saturated fat without adverse effects on cardiovascular disease is inconsistent with a large body of epidemiologic and intervention data.(27)
Is Reconciliation Possible?
How can these two camps be reconciled? First, the low carbohydrate diet studies have been conducted in relatively small numbers of subjects for relatively short periods of time. It is possible that the effects of low carbohydrate diets on lipids may change over a period of time exceeding the study durations.
Second, it is likely that several factors determine the susceptibility of individuals to adverse lipid responses. It is possible that genetic susceptibility plays a significant role in determining the individual response, but at present there are no methods to identify such individuals. Therefore, it seems prudent to avoid diets at the extremes of high fat or high triglyceride intake in those individuals who demonstrate very high levels of total LDL cholesterol or triglycerides (respectively).
Third, the effect of diet composition may be minimized by negative energy balance. This principle is best illustrated by the effects of very low-calorie diets on blood glucose control in diabetics. If the change in blood sugar is examined relative to the duration of the very low-calorie diet, it becomes clear that reducing intake, even without significant weight loss, has a powerful effect on blood sugar. Within a week of consuming a very low-calorie diet, blood sugar decreases almost 50% of the total decrease observed following 16 weeks of dieting.(28)
Conclusions
The macronutrient content of the diet affects both sides of the energy equation, intake and expenditure. It appears that the most meaningful effects are observed on food intake, while the effects on energy expenditure and nutrient partitioning may have a less significant role on body weight regulation. Diets comprised of very low carbohydrate content may be effective for some, but the long-term efficacy remains questionable. In addition, while no overall ill effects on blood lipids or other cardiovascular risk factors have thus far been observed, more studies are warranted to better assess the effects of long-term use of very low carbohydrate diets on metabolic diseases and cardiovascular risk factors. Therefore, further research is needed to better define which dietary macronutrient composition is optimal.