Course Authors
Leon Charkoudian, M.D., and Joshua L. Dunaief, M.D., Ph.D.
Dr. Charkoudian is a resident and Dr. Dunaief is Assistant Professor of Ophthalmology, F.M. Kirby Center for Molecular Ophthalmology, Scheie Eye Institute, University of Pennsylvania, Philadelphia, PA.
Within the past 12 months, Drs. Charkoudian and Dunaief report no conflicts of interest.
Albert Einstein College of Medicine, CCME staff and interMDnet staff have nothing to disclose.
Estimated course time: 1 hour(s).

Albert Einstein College of Medicine – Montefiore Medical Center designates this enduring material activity for a maximum of 1.0 AMA PRA Category 1 Credit(s)™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
In support of improving patient care, this activity has been planned and implemented by Albert Einstein College of Medicine-Montefiore Medical Center and InterMDnet. Albert Einstein College of Medicine – Montefiore Medical Center is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Upon completion of this Cyberounds®, you should be able to:
Describe the basics and natural history of age-related macular degeneration
Identify who should be screened for age-related macular degeneration and when a patient should be referred for further evaluation
Discuss the treatments available for age-related macular degeneration.
Age-related macular degeneration (AMD), also known as age-related maculopathy, is a potentially progressive macular disease that is the leading cause of adult visual impairment and blindness in industrialized nations. It typically affects whites over the age of 55. Characteristic ophthalmoscopic features of the disease are the development of discrete macular lesions (drusen), macular pigmentary changes, macular atrophy and neovascularization of the underlying choroidal vasculature with exudation. Diagnosis and treatment are highly specialized and involve consultation with a retinal specialist.
Epidemiology
It is estimated that approximately 7 million individuals in the U.S. have intermediate AMD and 1.75 million have advanced AMD.(1) Although most individuals (approximately 80%) have the non-neovascular form (also known as the "dry" form), the neovascular form (or "wet" form) accounts for the majority of the cases with severe visual loss.(2) One study revealed that the incidence of disease and its associated features increase with age and are low in individuals younger than 55 years.(3) Multiple studies have shown that advanced AMD is significantly more prevalent in white compared to African-American or Hispanic populations but prevalence may also be relatively higher in Asian populations.(4)(5)(6)
Pathophysiology
The cause of AMD is unclear but it likely results from a combination of oxidative damage, microvascular disease and genetic predisposition.
Multiple studies have shown that oxidative damage is associated with drusen formation and retinal pigment epithelial cell degeneration.(7)(8)(9)(10) The finding that antioxidant vitamins and minerals slow disease progression supports these proposals.(11) Deposition of extracellular material ("drusen") along the inner aspect of Bruch's membrane -- the membrane separating the retina from the choroid -- is thought to lead to chronic inflammation and oxidative stress. Oxidative damage accumulates over time and is consistent with the age-related nature of the disease, as well as the increase in number and size of drusen. It is thought that metabolic stress leads to cellular damage and secondary loss of adjacent photoreceptors. Oxidative stress may also provoke increased production of vascular endothelial growth factor by retinal cells, which is proposed to be the stimulus for neovascularization of the choroidal vasculature.
Some microvascular diseases, such as hypertension and atherosclerotic heart disease, have been associated with increased prevalence of AMD, which may suggest that these conditions affect cellular protection from oxidative damage. However, data are conflicting.(12)(13)(14)
Epidemiologic surveys have found higher rates of disease within families and in twins compared with the general population, which indicate a genetic predisposition to the disease.(15)(16)
Polymorphisms that affect complement systems -- biochemical pathways that have roles in inflammatory processes -- probably contribute to overactivity of the immune system, which subsequently leads to inflammation and cellular damage. Individuals homozygous for the 402H complement factor H polymorphism are at up to seven times increased risk of developing AMD compared to the general population.(17)(18)(19)(20)
Risk Factors
The most widely identified risk factors are:
Increasing Age
Large epidemiologic studies show that incidence and prevalence of disease and associated features increase with age. Prevalence of AMD in individuals aged 75-85 years was found to be more than three times that of individuals aged 43 to 54 years.(4)
White Ethnicity
Advanced disease is significantly more prevalent in white compared with African-American or Hispanic populations. Prevalence may also be relatively higher in Asian populations.(4)(5)(6)
Smoking
Heavy smoking is associated with an increased risk of development of disease that is two times that of not smoking.(21)
Clinical data show a dose-related response, with increasing odds of developing advanced AMD directly related to number of pack-years smoked; this association holds true for all forms of advanced AMD (choroidal neovascularization and central geographic atrophy).
Cessation of smoking reduces the odds of developing advanced AMD.
Complement Factor Polymorphisms
Polymorphisms in complement factor H (CFH) and other complement factors have been shown to significantly increase the risk of disease.(17)(18)(20)
The CFH gene is located on chromosome 1 and this region has been linked in family studies to AMD.(19)
The association between CFH polymorphism and increased risk of disease is particularly strong for smokers.(22)
Hypertension
Hypertension is associated with increased prevalence of AMD and risk of disease progression but results are conflicting.(12)(13)(14)
Atherosclerotic Disease
Atherosclerotic cardiovascular disease is associated with increased prevalence of AMD but results are conflicting.(12)(13)(14)
Other potential risk factors include low dietary intake of antioxidants, higher dietary intake of saturated fats and cholesterol, and sunlight exposure but studies on these factors are conflicting.(23)(24)(25)(26)(27)(28)(29)(30)(31)(32)
Protective Factors
No primary prevention strategies have been proven to be effective in preventing development of disease. Raising dietary intake of antioxidants and avoiding smoking are suggested to be beneficial. Antioxidant vitamin supplementation can decrease the risk of progression from mild to advanced disease (see Treatment section below) but has no proven role in protection against development of mild disease. Routine dilated eye exams are recommended by national organizations and can be useful to screen for earlier stages of the disease.(33)(34) Established guidelines recommend regular dilated eye examinations every 2-4 years for people aged 40-64 years and every 1-2 years for persons aged 65 years or older.
Diagnosis
A directed history and dilated fundus examination by an ophthalmologist are typically sufficient to diagnose AMD. Ancillary studies may be used as indicated to differentiate severity of disease and to plan treatment strategies.
Patients are typically elderly, white and may have a history of smoking but this last feature is often absent. In the early stages of the disease, patients are frequently asymptomatic and may have no visual complaints or very mild distortion. A patient presenting with the neovascular form of the disease may report a sudden-onset distortion in the central visual field of one eye. Since early disease is often asymptomatic, routine dilated eye exams are recommended.
Dilated fundus examination identifies characteristics of disease and determines stage of disease, and is carried out by an ophthalmologist. The most widely accepted disease criterion is the Age-Related Eye Disease Study Group (AREDS) Classification:(11)

Table 1. Age-Related Eye Disease Study Group (AREDS) Classification.
| AREDS Category | Description |
|---|---|
| 1 (No AMD) | No or a few small (<63 micrometers in diameter) drusen. |
| 2 (Early AMD) | Many small drusen or a few intermediate-sized (63-124 micrometers in diameter) drusen or macular pigmentary changes. (Figure 1) |
| 3 (Intermediate AMD) | Extensive intermediate drusen or at least one large (≥125 micrometers) drusen, or geographic atrophy not involving the foveal center. (Figure 2) |
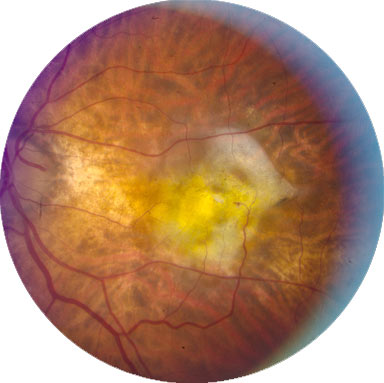
| 4 (Advanced AMD) | Choroidal neovascularization (Figure 3), geographic atrophy involving the foveal center (Figure 4) or evidence for neovascular maculopathy (subretinal hemorrhage, serous retinal or retinal pigmented epithelial detachments, lipid exudates or fibrovascular scar. (Figure 5) |

Figure 1. Early Age-Related Macular Degeneration (AREDS Category 2).
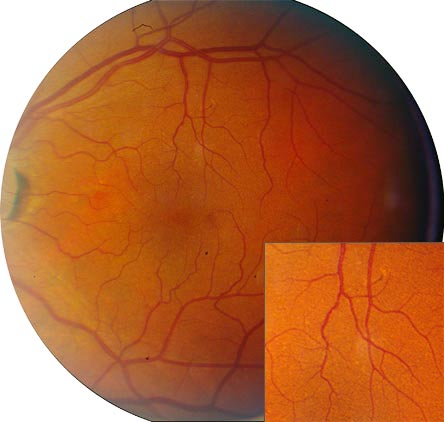

Figure 2. Intermediate Age-Related Macular Degeneration (AREDS Category 3).
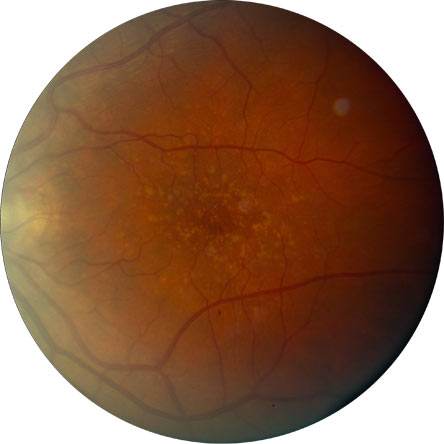

Figure 3. Advanced Age-Related Macular Degeneration (AREDS Category 4) with Choroidal Neovascularization and Exudation.
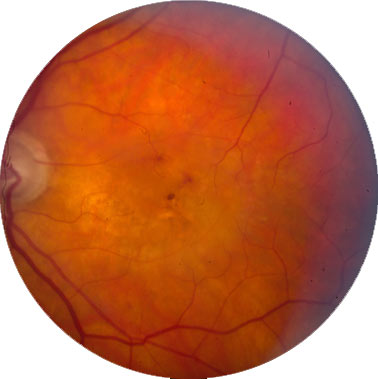

Figure 4. Advanced Age-Related Macular Degeneration (AREDS Category 4) with Central Geographic Atrophy.
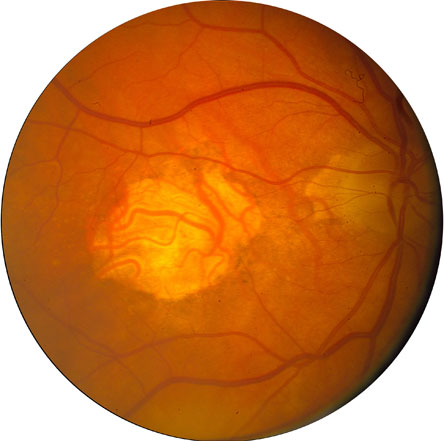

Figure 5. Advanced AMD (AREDS Category 4) with a Subfoveal Fibrous Scar Resulting from Choroidal Neovascularization.

Imaging is used as an adjunct to clinical examination. The most commonly utilized methods of diagnostic imaging are:

Table 2. Common Diagnostic Imaging Methods.
| Imaging Modality | Description |
|---|---|
| Fluorescein angiography | A vegetable-based dye is instilled intravenously and photographs are taken of the dye in the retinal circulation. Active choroidal neovascularization is detected as areas of expanding hyperfluorescence, indicating leakage. |
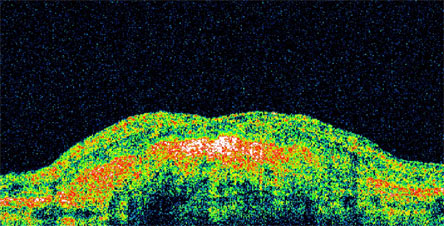
| Optical coherence tomography | Computerized imaging technique based on low-coherence interferometry. A cross-section of the retina is obtained. The presence of intra-retinal or sub-retinal fluid may be the result of active choroidal neovascularization with exudation. Loss of normal retinal architecture may represent atrophic areas. Long-standing disease may result in a dense hyperreflective scar. (Figure 6) |

Figure 6. Optical Coherence Tomography Image Showing a Dense Hyperreflective Scar.

Differential Diagnosis
Basal Laminar Drusen
- Multiple, discrete, often calcified drusen, symmetric in both eyes.
- Commonly seen in younger patients and non-white ethnicities, as opposed to typical AMD.
- Uncommonly associated with choroidal neovascularization, as opposed to typical AMD.
Myopic Degeneration
- Present in patients with high myopia (nearsightedness).
- May be associated with macular pigmentary changes and choroidal neovascularization similar to AMD.
- Differentiated on the lack of drusen and typical features of high myopia: tilted nerve head and posterior staphyloma (protrusion).
- Commonly seen in younger patients and non-white ethnicities, as opposed to typical AMD.
Ocular Histoplasmosis Syndrome
- High incidence in people residing in the Mississippi River Valley and nearby areas.
- May be associated with macular pigmentary changes and choroidal neovascularization similar to AMD.
- Differentiated on the lack of drusen and typical features of ocular histoplasmosis syndrome: peripapillary atrophy and punched-out chorioretinal scars in the macula and periphery.
- Commonly seen in younger patients and non-white ethnicities, as opposed to typical AMD.
Angioid Streaks
- Characterized by the presence of angioid streaks (yellow-orange streaks emanating from the optic nerve head) on funduscopic examination.
- Are associated with Ehler-Danlos syndrome, Paget's disease, pseudoxanthoma elasticum and sickle cell disease.
- May be associated with macular pigmentary changes and choroidal neovascularization similar to AMD.
- Differentiated on the lack of drusen and typical features of angioid streaks.
- Commonly seen in younger patients and non-white ethnicities, as opposed to typical AMD.
Traumatic Choroidal Neovascularization
- Trauma may be associated with rupture of Bruch's membrane (the layer separating the retina from the choroid) with possible choroidal neovascularization.
- Drusen are typically not present. Other evidence for trauma may be seen.
- Usually unilateral.
- Commonly seen in younger patients and non-white ethnicities, as opposed to typical AMD.
Idiopathic Choroidal Neovascularization
- Choroidal neovascularization without identifiable cause.
- Drusen typically not present.
- Usually unilateral.
- Commonly seen in younger patients and non-white ethnicities, as opposed to typical AMD.
Treatment
All patients suspected to have visual loss, particularly from AMD, should be referred to an ophthalmologist as soon as possible. Evaluation by a retinal-specialized ophthalmologist is recommended at any point in the disease process but is particularly necessary for any patient in whom AREDS category 3 or greater is reached in one eye; in whom subjective visual changes are experienced; or if diagnosis is uncertain and/or atypical features are present.
tation with select antioxidants has been shown to slow the progression of intermediate AMD to advanced AMD.
Supplementation with select antioxidants (vitamin C 500 mg; vitamin E 400 IU; beta carotene 15 mg (25,000 IU); zinc oxide 80 mg; cupric oxide 2 mg) has been shown to slow the progression of intermediate AMD to advanced AMD and associated visual loss.(11) The micronutrients are recommended for any patient with intermediate AMD or any individual with unilateral advanced AMD and a fellow eye at risk for progression. There was not a statistically significant effect on patients with no or early AMD. Commercially available products are available that combine the micronutrients as twice-daily supplements. Intake of high doses of beta carotene in current or recent ex-smokers may increase the risk of lung cancer and, consequently, smokers are advised to take an antioxidant supplement that does not contain beta carotene.(35)(36)
Treatment is based on the level of disease at presentation:

Table 3. AREDS Category v. Treatment.
| AREDS Category | Descriptione |
|---|---|
| 1 (No AMD) | Careful observation with risk factor modification. |
| 2 (Early AMD) | Careful observation with risk factor modification. Fundus photographs are taken to follow the disease process. |
| 3 (Intermediate AMD) | Careful observation with risk factor modification. Fundus photographs are taken to follow the disease process. Micronutrient supplementation as recommended by the AREDS (see below). |
| 4 (Advanced AMD without active choroidal neovascularization) | Careful observation with risk factor modification. Fundus photographs are taken to follow the disease process. Micronutrient supplementation as recommended by the AREDS (see below) if the fellow eye is not advanced as well. |
| 4 (Advanced AMD with active choroidal neovascularization) | Treatment is dependent on the size, subtype and location of the choroidal neovascularization -- see the following section. Fundus photographs are taken to follow the disease process. Micronutrient supplementation as recommended by the AREDS (see below) if the fellow eye is not advanced as well. |

At this time, there is no proven treatment for early disease (AREDS categories 1 and 2) and management is based on careful observation with risk factor modification.
For patients with intermediate stage disease (AREDS category 3), risk factor modification (i.e., smoking cessation, dietary modification and atherosclerotic risk factor modification) is coupled with AREDS formula antioxidant supplementation.
For patients with advanced stage disease (AREDS category 4), treatment is indicated if active choroidal neovascularization is present. Treatment for choroidal neovascularization is highly specialized and is carried out by an ophthalmologist specializing in diseases of the retina. AREDS formula antioxidant supplementation is recommended for persons with unilateral advanced AMD with a second eye at risk of progression. Patients with AREDS category 4 disease without active choroidal neovascularization are followed closely with serial examinations and are instructed to report immediately any visual changes.
Treatments for active choroidal neovascularization depend on the size, subtype and location of the choroidal neovascularization. They include:
Thermal Laser Photocoagulation
- Direct laser application to the area of choroidal neovascularization.
- May be preferred for choroidal neovascularization outside of the fovea.(37)(38)
- Treatment is given as soon as possible after identification of active choroidal neovascularization to prevent irreversible retinal damage. Treatment response is monitored closely with fluorescein angiography. Retreatments may be necessary.
- Treatment of extrafoveal and juxtafoveal choroidal neovascularization by laser photocoagulation has been shown to significantly decrease the risk of severe visual loss but recurrence is common.(37)(38)(39)
Photodynamic Therapy
- Involves intravenous injection of a photosensitizer (a chemical compound that creates oxidative damage when excited by a specific light wavelength) and the application of a beam of light at the specific activating wavelength to the area of choroidal neovascularization.
- May be used for choroidal neovascularization under the fovea.(40)(41)(42)(43) Considered second-line to anti-VEGF agents as sole therapy for active subfoveal choroidal neovascularization because of comparatively lower efficacy. Currently used on an experimental basis in combination with VEGF inhibitors.(44)(45)(46)(47) Retreatments may be necessary.
- Patients are instructed to avoid sunlight and cover all exposed skin when outside for the first five days following therapy to avoid activation of the photosensitizer within the skin and a burn-like reaction.
- Treatment of subfoveal choroidal neovascularization by photodynamic therapy reduces the rates of vision loss, but most patients still experience a decline in vision.(40)(41)(42)(43)
Intravitreal VEGF Inhibitors
- Direct injection into the vitreous of an inhibitor of vascular endothelial growth factor (VEGF).
- May be used for choroidal neovascularization under the fovea.(44)(45)(46)(47)
- Examples include pegaptanib, ranibizumab and bevacizumab. Studies have shown ranibizumab and bevacizumab to be more effective when compared with pegaptanib.
- Treatment is given as soon as possible after identification of choroidal neovascularization activity to prevent further retinal damage. Effects are relatively short-lived and therapy typically involves a series of injections (often monthly) depending on patient response. Treatment response is monitored closely with optical coherence tomography and fluorescein angiography.
- Treatment is associated with a small risk of endophthalmitis (infection within the eye), retinal detachment or traumatic lens injury. Risks of these complications are approximately 1%, <1%, and <1%, respectively. Patients are made aware of signs indicative of endophthalmitis (pain, decreased vision, significant light sensitivity and/or increasing redness of the eye) and retinal detachment (new floaters, flashing lights and/or obscured visual field).
- Treatment of choroidal neovascularization by intravitreal injection of anti-VEGF agents (ranibizumab or bevacizumab) results in improvement in vision in up to a third of patients and stabilization of vision in up to 95% of patients.(45)(46)(47)(48) Intravitreal injection of pegaptanib is considered second-line because of comparatively lower efficacy.(44)
Natural History
Numerous studies have revealed the natural history of AMD without treatment:

Table 4. Level of Disease v. Natural History.
| Level of Disease | Natural History |
|---|---|
| AREDS Category 2 (early AMD) | Typically, visual acuity remains unaffected unless disease progression occurs. Patients have a 1.3% risk over 5 years of progressing to advanced AMD.(11) |
| AREDS Category 3 (intermediate AMD) | Typically, visual acuity remains unaffected unless disease progression occurs. Patients have an 18% risk over 5 years of progressing to advanced AMD.(11) |
| AREDS Category 4 (advanced AMD) | Patients with unilateral disease have a 43% chance over 5 years of developing advanced AMD in the other eye.(11) Untreated, advanced AMD will result in significant visual loss (doubling of the visual angle or worse) in over half of patients over several years.(37)(38)(39)(48) |

Resources
- Foundation for Fighting Blindness: Macular Degeneration - http://www.blindness.org/MacularDegeneration/
- MedlinePlus: Macular Degeneration - http://www.nlm.nih.gov/medlineplus/maculardegeneration.html
- National Eye Institute: Macular Degeneration - http://www.nei.nih.gov/health/maculardegen/armd_facts.asp
Guidelines
Age-Related Macular Preferred Practice Pattern. American Academy of Ophthalmology. 09/2006. http://www.aao.org/education/guidelines/ppp/amd_new.cfm - Non-algorithmic summary of the diagnosis and treatment of AMD.
Case Presentation
A 75-year-old woman presents with new-onset central distortion in one eye. Vision is 20/80 in the involved eye, which reportedly had near-normal vision as recently as a few weeks ago. The involved eye is normal in external appearance.
The new-onset subacute central distortion in one eye is highly suggestive of advanced age-related macular degeneration from new choroidal neovascularization with exudation. For this reason, patients with earlier stages of macular degeneration (answer C), for which significant visual loss does not typically occur, are asked to screen each eye daily for new areas of distortion. Cataracts and glaucoma typically result in visual loss over months to years, not days to weeks; acute angle-closure glaucoma may result in a rapid decline in vision but the eye is usually red in external appearance and painful.
Dilated funduscopic examination of the involved eye reveals an area of subretinal fluid and hemorrhage with surrounding lipid exudate in the central macula (Figure 7; advanced age-related macular degeneration). Examination of the uninvolved eye reveals extensive yellow-white retinal lesions (drusen), macular pigmentary changes and areas of atrophy (Figure 8; intermediate age-related macular degeneration).

Figure 7. Fundus Photograph of the Right Eye.
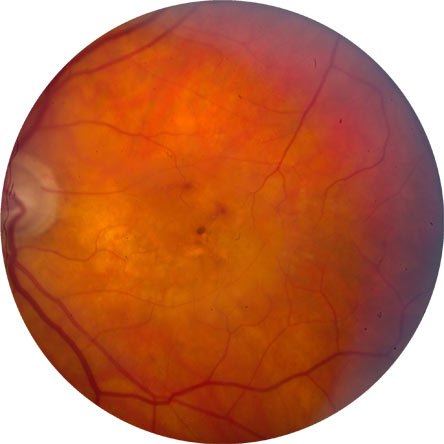
Shows fluid and hemorrhage in the macula ("wet" macular degeneration).

Figure 8. Fundus Photograph of the Fellow Eye.
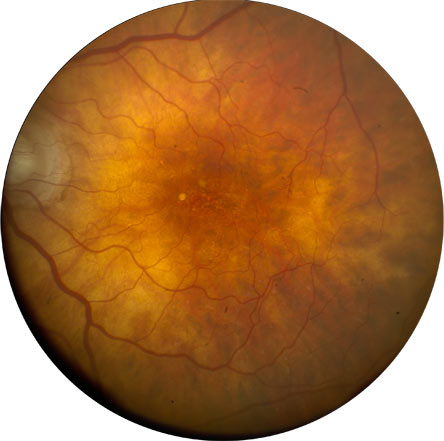
Shows drusen and pigmentary changes but no fluid or hemorrhage ("dry" macular degeneration).

The treatment of active choroidal neovascularization is dependent on the size and location of the disease. Determination of these features is done by fluorescein angiography, and optical coherence tomography is performed to confirm exudation. Refraction is not indicated and is done to determine refractive errors in patients without other active conditions.
Fluorescein angiography of the involved eye reveals a central area of expanding hyperfluorescence consistent with leakage from neovascular choroidal blood vessels (Figure 9). Optical coherence tomography confirms the presence of subretinal and intraretinal fluid secondary to exudation from the neovascular vessels (Figure 10).

Figure 9. Fluorescein Angiographic Image of the Right Eye.
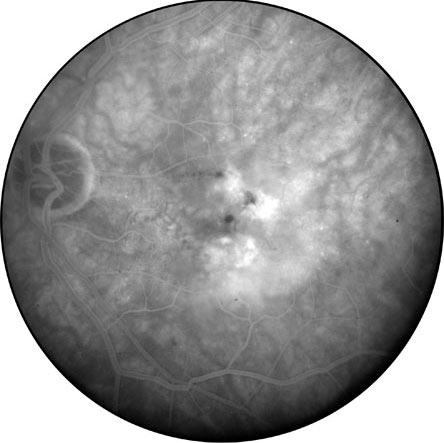
Shows areas of hyperfluorescence corresponding to macular leakage from choroidal neovascular blood vessels.

Figure 10. Optical Coherence Tomography Image of the Macula of the Same Eye.
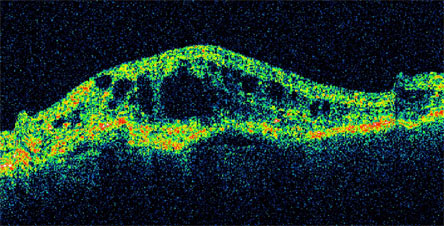
Shows areas of intra- and sub-retinal fluid from leaking neovascular choroidal vessels.

Active choroidal neovascularization may be treated with anti-VEGF intravitreal injections, photodynamic therapy, or laser photocoagulation, depending on the size and location of the choroidal neovascularization. Patients with unilateral advanced age-related macular degeneration with a fellow eye at risk, such as this patient, should take micronutrients as described by the AREDS to slow the risk of progression in the fellow eye. Micronutrient supplementation should not be ignored, as it is the only proven treatment for the intermediate stage of the disease before visual loss typically occurs.
Smoking, hypertension and increased dietary intake of saturated fats and cholesterol have all been linked to increased risk of AMD. Data on sunlight exposure are conflicting and this cannot be definitely linked as a risk factor.
National organizations recommend screening for asymptomatic healthy patients every 2-4 years for people aged 40-64 years and every 1-2 years for persons aged 65 years or older. Note that patients with a history of eye disease, relevant systemic diseases (such as diabetes mellitus) or strong family history are screened earlier and more frequently.
National organizations recommend screening for asymptomatic healthy patients every 2-4 years for people aged 40-64 years and every 1-2 years for persons aged 65 years or older. Note that patients with a history of eye disease, relevant systemic diseases (such as diabetes mellitus) or strong family history are screened earlier and more frequently.