Course Authors
Ali H. Shakir, M.D., Richard W. Smalling, M.D., Ph.D., and Rebecca R. Hung, M.D.
Release Date: 12/01/2003
Upon completion of this Cyberounds®, you should be able to:
List the physiologic properties of B-type natriuretic peptide (BNP) and NT-proBNP
Describe the utility and limitations of measuring plasma BNP and NT-proBNP levels in the diagnosis and management of congestive heart failure
Discuss the role of recombinant BNP (nesiritide) in the treatment of decompensated heart failure.
Congestive heart failure remains a major clinical challenge that spans all domains of medicine. Please welcome Dr. Ali Shakir, one of our Cardiovascular Medicine Fellows and Dr. Rebecca Hung to Cyberounds®. Dr. Hung recently joined our faculty from the Massachusetts General Hospital and Harvard Medical School where she trained in Cardiovascular Medicine with a special emphasis on the treatment of heart failure. Drs. Shakir and Hung have prepared an informative and up-to-date review of new methodologies for the accurate diagnosis of and optimal treatment for acute congestive heart failure.
Richard Smalling, M.D., Ph.D.
Cardiovascular Medicine Editor
Congestive heart failure is a leading cause of mortality and hospitalization, particularly in the elderly.(1) Despite the frequency of this diagnosis, the clinical assessment of congestive heart failure continues to be challenging, particularly in patients with chronic heart failure and those with multiple comorbidities such as obesity and underlying lung disease. The signs and symptoms of heart failure are neither adequately specific nor sensitive for accurate diagnosis.(2) Even the presence of left ventricular dysfunction by echocardiography is not synonymous with clinical heart failure.
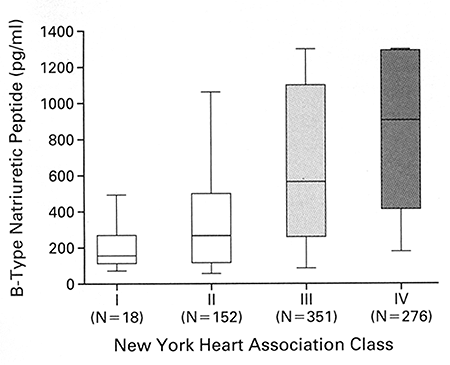
Work over the past decade has demonstrated the utility of measuring plasma B-type natriuretic peptide (BNP) or its leader peptide sequence, NT-proBNP, to serve as a marker of heart failure that is independent of etiology(3),(4) BNP and NT-proBNP levels correlate with New York Heart Association class symptoms(5),(6) (Figure 1).
They are elevated in ischemic and non-ischemic cardiomyopathies, aortic stenosis, left ventricular hypertrophy, hypertrophic cardiomyopathy, right ventricular failure as well as asymptomatic left ventricular dysfunction. Both BNP and NT-proBNP levels fall favorably in response to aggressive treatment of heart failure(6) and mirror falls in intracardiac filling pressures.(7) Recombinant human BNP (nesiritide) has been shown to have a therapeutic role in the management of decompensated heart failure and should occupy a unique niche amongst the therapeutic options available to the acutely ill patient.(8) This review summarizes recent advances in the utilization of BNP and NT-proBNP for diagnosis and recombinant BNP for treatment.
Case Presentation
A 70-year-old African American female with a history of peripheral vascular disease, hypertension, sick sinus syndrome and diabetes presented to our institution with one week of progressive right upper quadrant pain and weight gain. She reported dyspnea with minimal exertion.
Physical exam demonstrated a tired appearing African American female. Blood pressure was 130/70 mm Hg; pulse was 60 beats per minute; respirations were 16 breaths per minute; weight was 150 pounds. Jugular venous pressure was estimated at greater than 14 cm of water. Lungs were clear. Heart exam demonstrated a regular rate with a prominent right ventricular impulse and a loud P2. The apical impulse was displaced laterally. A III/VI holosystolic murmur of tricuspid regurgitation was audible along the left lower sternal border. Pulsatile hepatomegaly was present along with hepatojugular reflux. Extremities were cool with 4+ pitting edema to the thighs.
Laboratory data were notable for a potassium 3.1 meq/L, BUN 28 mg/dL and creatinine of 1.4 mg/dL. NT-proBNP was >29,000 pg/mL. BNP was >5000 pg/mL, the maximum linear range of the test. Transaminases were mildly elevated. Chest X-ray revealed mild cardiomegaly without pulmonary congestion. Echocardiogram demonstrated a non-dilated left ventricle with an ejection fraction of 23% with mild concentric left ventricular hypertrophy. The right ventricle was markedly dilated with severe tricuspid regurgitation. Biatrial enlargement was present, as was a restrictive pattern of mitral inflow consistent with elevated left atrial pressure.
During the initial 48 hours of hospitalization, the patient was unresponsive to escalating doses of intravenous furosemide (up to 160 mg) in addition to metolazone. Creatinine rose to 2.5 mg/dL. On hospital day 3, intravenous nesiritide was initiated at a fixed dose of 0.01 mcg/kg/min following a 2 mcg/kg bolus. Within 6 hours, urine output increased and over the next 72 hours, effective diuresis of 4-5 liters of urine per 24-hour period was achieved with only a single daily dose of intravenous furosemide of 40 mg. Serum creatinine stabilized at 1.9 mg/dL. With a net diuresis of 30 lbs, the patient's symptoms of hepatic congestion resolved. The patient's dyspnea also improved.
This case, not atypical of the patients seen in our institution, illustrates the utility of B-type natriuretic peptide in the diagnosis and management of decompensated heart failure.
Physiology of B-Type Natriuretic Peptide
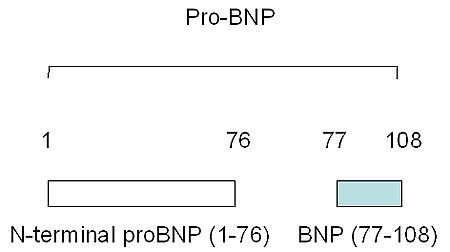
B-type (brain) natriuretic peptide (BNP) is a member of a family of vasoactive hormones involved in the regulation of blood pressure and volume homeostasis. Although originally isolated from porcine brain, it is primarily synthesized in ventricular myocardium in response to myocyte stretch.(9) It is a 32 amino acid polypeptide containing a 17 amino acid ring structure common to all natriuretic peptides (Figure 2).
Upper panel shows active BNP as the carboxy terminal polypeptide (amino acid 77-108) of the prohormone, Pro-BNP. The N-terminal fragment NT-ProBNP (amino acid 1-76) is biologically inactive.
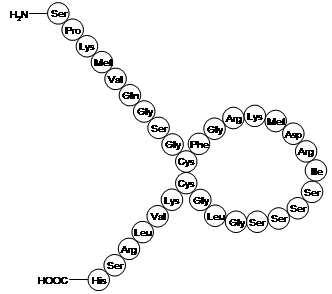
Bottom panel shows amino acid sequence and cyclic structure of BNP.
BNP binds its receptor on the surface of target cells and leads to second messenger generation via particulate guanylate cyclase. BNP release is regulated at the gene transcription level and thought to be a more reliable and stable indicator of ventricular volume status than ANP (atrial natriuretic peptide), a similar hormone expressed primarily in the atria.(10) CNP, a related peptide, is produced in vascular endothelium. Both ANP and BNP have beneficial effects on the failing heart (Table 1).
Table 1. Beneficial Effects of Natriuretic Peptides on the Failing Heart.
|
BNP affects balanced vasodilation of smooth muscle, able to simultaneously reduce both preload and afterload as a combined arterial and venous vasodilator. As a result, both systemic blood pressure and ventricular preload are affected by BNP. BNP inhibits the sympathetic nervous system and the renin-angiotensin axis, thus modulating the synthesis of hemodynamically detrimental vasoconstrictors such as catecholamines, renin, aldosterone and endothelin-1.
BNP has lusitropic (relaxing) properties on the myocardium that may play a role in the treatment of diastolic dysfunction. BNP may also have antiproliferative and antifibrotic effects. In the kidney, sodium and water excretion is enhanced with preservation of glomerular filtration. With this combination of properties, endogenous BNP plays a role in the compensatory mechanisms of the failing heart. It is precisely this compensatory capacity that positions BNP as a unique candidate both as a marker of and a therapeutic target in congestive heart failure.
The precursor peptide, pro-BNP, is a 108 amino acid polypeptide that is secreted and rapidly cleaved enzymatically to the active BNP and the inactive NT-proBNP (N-terminal proBNP) [Figure 2]. BNP is cleared by receptor mediated endocytosis and enzymatic degradation by neutral endopeptidases. The half-life of BNP is reported to be short, approximately 20 minutes, and is thus cleared within 2 hours (5 half-lives). A change in level of BNP thus reflects an acute change in a given patient's clinical status. NT-proBNP has a longer half-life of 1-2 hours giving a broader measuring range.
In patients with normal renal function, BNP and NT-proBNP levels parallel each other well, but in renal failure both are less accurate. It is thought that both BNP and NT-proBNP are elevated in patients with renal disease partly due to increased intravascular volumes. The majority of studies that establish BNP and/or NT-proBNP as screening tools excluded patients with significant renal dysfunction and the interpretation of either of these levels in renal failure has not been firmly established. It should be noted that, in the therapeutic use of nesiritide, however, there is no dose adjustment for renal dysfunction.
Three assays(11) are currently commercially available for clinical use to measure BNP or its surrogate NT-proBNP. (It is important for the practitioner to be aware that when ordering a "BNP" level, the assay may, at times, actually be the assay for NT-proBNP and the reference levels will be different.) NT-proBNP has been demonstrated to have longer in vitro stability compared with BNP (72 versus 6 hours), and has been successfully used for optimizing an outpatient regimen for heart failure. In addition, NT-proBNP can be used to monitor clinical response during acute nesiritide therapy as it is structurally distinct from the active agent.
A value of 100 pg/mL for BNP is considered normal.(12) Because of NT-proBNP's broader measuring range, normal values show greater age dependence with 125 and 450 pg/mL set as cutoffs for less than 75 and greater than 75 years of age, respectively.(12) These changes are thought to reflect diastolic dysfunction in the aging myocardium. Regardless of the choice of assay, one needs to be aware of the differences in normal values and dynamic range of the two assays when interpreting the results and to be cautious about comparing the results obtained by the two methods with different performance characteristics.(12)
Factors other than heart failure affect BNP levels.(13) Baseline values must take into consideration the age and gender of the patient. BNP levels increase with age, although it is not clear how much of this trend merely reflects the higher prevalence of hypertension, left ventricular hypertrophy and undiagnosed diastolic dysfunction in the older population. Women tend to have slightly higher levels than men. BNP levels are mildly elevated in renal failure but do decline following dialysis, reflecting to some extent the resolution of volume overload rather than true underlying renal dysfunction. Hepatic dysfunction and cirrhosis are also associated with rising BNP and NT-proBNP, although the changes are believed to reflect the hemodynamic changes of cirrhotic physiology.(14) BNP levels inversely correlate with body mass index. Given the confounding variables, a single measurement of BNP without clinical context is insufficient for making the diagnosis of heart failure.
Caveats to interpreting a BNP level for a given patient include the following. First, the presence of an elevated BNP level does not rule out the presence of concomitant diagnoses of pulmonary disease. Second, as BNP levels span a broad range of absolute values even for patients of a similar symptom class (NYHA), use of absolute BNP or NT-proBNP levels may not accurately reflect an individual patient's response to therapy. It may be more appropriate to follow the relative trend in BNP or NT-proBNP levels, pre- and post- initiation of treatment.
Diagnosis of Acute Congestive Heart Failure
In the recent multinational Breathing Not Properly trial, the Triage BNP test was used to measure plasma BNP levels in patients presenting to the emergency department with a chief complaint of shortness of breath.(5) Patients excluded from the study were those under age 18, those with dyspnea clearly not secondary to congestive heart failure, acute myocardial infarction, renal failure (creatinine greater than 2 mg/dl) and unstable angina. The diagnosis of heart failure was adjudicated by an independent panel of cardiologists.
Patients with congestive heart failure had higher levels of BNP than those without congestive heart failure. Patients with known systolic dysfunction, but who had an alternative etiology of dyspnea, had BNP levels that were intermediate to those with heart failure and non-cardiac dyspnea. The cutoff of 100 pg/ml showed a sensitivity of 82% and specificity of 95% for the diagnosis of heart failure. Although the mean BNP values between groups were significantly different, the range of BNP values between groups did overlap. Possible reasons for this overlap include some of the factors discussed earlier.
Patients with lung disease and concomitant right sided heart failure may also have elevated plasma BNP and NT-proBNP, although the values tend to be lower than those with left ventricular failure. Similarly, patients with pulmonary embolism have pressure overload of the right ventricle and may also have elevated BNP levels.(4) Most studies have excluded patients with renal failure so that generalizations regarding BNP level in those situations cannot be made. Together, these data suggest that while BNP level alone cannot be used to make a definitive diagnosis, it is a useful adjunct to support clinical decision making and improves the accuracy of diagnosis.
There are also instances when clinical heart failure may be associated with a normal BNP level, including obese patients with an elevated body mass index,(13) and patients with end stage heart failure who appear to have an inability to synthesize adequate BNP.(15) Thus, while a normal BNP level is useful to exclude the diagnosis of heart failure in patients with a low or intermediate pretest probability, false negative results need to be examined in clinical context when the suspicion for heart failure is high.
Screening Tool for Heart Failure
BNP has been proposed as a low cost screening tools for heart failure in lieu of echocardiography.(16) It would potentially identify asymptomatic left ventricular dysfunction that may benefit from treatments such as angiotensin converting enzyme inhibitors and beta blockers that may prevent the progression to symptomatic heart failure. Screening could be targeted towards subgroups at risk for left ventricular dysfunction, such as patients with diabetes, hypertension, coronary artery disease with history of myocardial infarction or patients undergoing chemotherapy with cardiotoxic agents such as the anthracyclines.
Within a group of 400 patients referred for echocardiography, BNP levels were able to detect the presence of left ventricular dysfunction on echocardiography(16) but it was unable to distinguish between systolic and diastolic dysfunction. Conversely, in a community-based cohort,(17) the Framingham Heart Study group found that BNP did not assist in identifying patients with asymptomatic left ventricular systolic dysfunction or left ventricular hypertrophy. In those patients pre-selected for echocardiography based on clinical symptoms or risk factors warranting evaluation of ventricular function, a normal BNP level may obviate the need for echocardiography. However, current data do not support the use of BNP for screening for left ventricular dysfunction in the general population. It has been reported that NT-proBNP is similarly a predictor of echocardiographic left ventricular dysfunction.(18)
Diagnosis of Diastolic Heart Failure
Doppler echocardiography has traditionally been used to examine left ventricular diastolic dysfunction. In a study done by Lubien et al. comparing BNP levels and Doppler velocity recordings,(19) BNP levels were noted to be increased in patients with diastolic dysfunction. A cutoff value of 62 pg/ml had a sensitivity of 85% and specificity of 83% of diagnosing diastolic dysfunction. BNP levels were progressively higher with worsening severity of diastolic dysfunction and were highest in patients with E/A ratios >1.5 and in those patients with deceleration times less than 160 seconds consistent with a restrictive filling pattern by echocardiography and severe diastolic dysfunction. However, given significant overlap, BNP levels were unable to distinguish among the various intermediate echocardiographic grades of diastolic dysfunction. Similar to assessing systolic function, BNP levels can be used to identify, but not further categorize, diastolic dysfunction.
BNP and NT-proBNP Guided Treatment of Congestive Heart Failure
BNP levels correlate with volume status changes of patients undergoing hemodynamic monitoring. In a small study of patients admitted with heart failure,(7) Kazenegra et al. reported that pulmonary capillary wedge pressures and clinical improvement correlated with falling BNP levels. Larger studies may be able to better assess the potential of the BNP assay to replace invasive pulmonary artery catheterization as an indicator of pulmonary capillary wedge pressure. Although BNP levels clinically remain promising as a tool to measure volume status, further guidelines are needed to determine when BNP can appropriately replace invasive monitoring.
Providing optimal medical management for chronic heart failure in the outpatient setting can prove challenging, as the clinical exam often underestimates filling pressures.(2) To examine the incremental benefit of following serial NT-proBNP levels in the outpatient titration of a medical regimen for heart failure, Troughton et al. compared medical management guided by NT-proBNP levels with that solely determined by clinical assessment.(20) Patients who were treated on the basis of NT-proBNP had a lower primary combined endpoint of cardiovascular death, hospital admission and outpatient heart failure. Of note, patients in the NT-proBNP group were placed on higher dosages of ACE inhibitors, diuretics and spironolactone. NT-proBNP guided treatment may allow the clinician to be more aggressive in targeting pharmacotherapy. Although only a single center study, these data illustrate the potential of the NT-proBNP assay to improve the outcomes in outpatient heart failure therapy.
BNP and NT-proBNP as Prognostic Markers
In heart failure with left ventricular dysfunction, higher BNP levels correlate with increased cardiovascular and all-cause mortality, independent of age, NYHA class, previous infarction or ejection fraction.(21) BNP level is an independent predictor of sudden cardiac death.(22) NT-proBNP is a similar independent predictor of adverse outcomes, including all cause mortality, need for urgent transplanatation and hospital admission.(23) While markedly elevated levels of BNP generally identify a higher risk subgroup, in a small observational study of patients with chronic heart failure, BNP levels were noted to decline in end stage heart failure, suggesting there are limitations to the compensatory capacity of BNP production.(15) Thus, declining BNP levels in patients that exhibit clinical deterioration may be a harbinger of impending pump failure, and may prompt the physician to initiate a discussion of end of life care.
At the completion of in-hospital treatment, a failure to reduce BNP level despite clinical improvement, is a predictor of the need for subsequent readmission(24) A favorable reduction in BNP in response to treatment carries a good prognosis for freedom from 30-day readmission for heart failure.(24) In contrast, despite clinical improvement in the initial admission, the patients who did not exhibit a decrease in their BNP levels had a higher incidence of readmission. Thus, while the presenting BNP level may be helpful in the initial diagnosis of heart failure, a follow-up BNP or NT-proBNP prior to hospital discharge may be a useful adjunct to clinical examination for the adequacy of therapy.
Following Acute Coronary Syndromes
Elevated BNP has been associated with a higher risk of adverse outcomes in patients enrolled in the TIMI-18 (Thrombolysis in Myocardial Infarction) study presenting with non-ST elevation acute coronary syndromes.(25) A BNP level of greater than 80 pg/mL was associated with an increased risk of early death at 7 days and later death at 6 months, independent of other clinical predictors, including cardiac troponin I and the presence of clinical heart failure. There was a 5-fold higher risk of developing new congestive heart failure at 30 days. BNP levels were not predicted by differences in an invasive versus non-invasive management strategy. NT-proBNP has similarly been found to be a predictor of risk in acute coronary syndromes.(26) BNP screening at the time of an acute coronary syndrome may help to identify a subgroup at risk for adverse events.(27) It remains unknown, however, at this time, if more aggressive medical therapy will alter the outcomes.
BNP for the Treatment of Heart Failure
Recombinant human BNP (nesiritide) is now commercially available for parenteral use in decompensated heart failure. The beneficial properties of endogenous BNP including balanced vasodilation, inhibition of the renin-angiotensin axis, natriuresis and diuresis while maintaining GFR, with the hemodynamic benefits of decreased pulmonary capillary wedge and pulmonary artery pressures as well as increased cardiac output, combine to make BNP an attractive candidate for the management of acutely decompensated heart failure. In an early trial examining the acute hemodynamic effects in a group of 103 patients with NYHA Class III and IV heart failure with ejection fractions less than 35%, baseline pulmonary capillary wedge pressure (PCWP) of 29 mm Hg and a mean cardiac index of 1.9 L/min, nesiritide resulted in a 30% decrease in PCWP by six hours accompanied by a significant increase in CI, without a change in heart rate.(28) The hemodynamic benefits, evident within one hour of initiating therapy were not, however, sustained beyond termination of the infusion.
The clinical efficacy of nesiritide was examined in a randomized double-blind, placebo-controlled trial of 127 patients, reported by Colucci, et al.(29) Entry criteria included the use of at least one intravenous agent in addition to diuretics. This study demonstrated a dose dependent decrease in PCWP, right atrial pressure, systemic vascular resistance and systolic blood pressure. There were statistically significant increases in cardiac index and 6-hour urine output. Diuretic requirements were reduced in the groups receiving nesiritide as compared to placebo.
In the Vasodilation in the Management of Acute Congestive Heart Failure (VMAC) trial,(8) 489 patients with decompensated heart failure who presented with dyspnea at rest were randomized to receive either intravenous nesiritide or nitroglycerin. The patients who received nesiritide (2 mcg/kg bolus, followed by 0.01 mcg/kg/min infusion with an option to titrate up to 0.03 mcg/kg/min) had a significant reduction in pulmonary capillary wedge pressure within 15 minutes, as compared with the nitroglycerin group. The difference persisted to 24 hours. Nesiritide initially increased cardiac output at 1 hour, but there were no significant differences in cardiac output by 3 hours. Doses of nesiritide above 0.01mcg/kg/min were associated with more frequent hypotension but no further significant improvement in hemodynamics.
Within the VMAC trial, a portion of the patients were treated without invasive hemodynamic monitoring, demonstrating the safety of nesiritide in the absence of pulmonary artery catheterization. Notably, the trial did not exclude patients with acute coronary syndromes and included a significant subset (15%) of patients with diastolic dysfunction and ejection fractions greater than 40%. No difference in symptomatic hypotension or systemic blood pressure was observed between the two therapies; however, more frequent headache was associated with nitroglycerin. Unlike nitroglycerin, tachyphylaxis was not seen with nesiritide, thus obviating the need for continuous dose titration. In the setting of invasive monitoring, nesiritide and nitroglycerin achieve similar hemodynamic effects, when the nitroglycerin dose was increased appropriately. In the absence of invasive monitoring, particularly given the inaccuracies associated with clinical assessment of filling pressures, nesiritide offers the advantage of fixed dosing with ease of administration and a reasonably predictable hemodynamic response.
In addition to nitroglycerin, nitroprusside has proven to be effective in the optimization of hemodynamics in decompensated heart failure under pulmonary artery catheter guidance.(30),(31) It is well tolerated, even in severely decompensated heart failure(31) with etiologies that include severe aortic stenosis with left ventricular dysfunction.(32) Patients receiving nitroprusside require invasive monitoring, as opposed to patients routinely receiving nesiritide.
In cases where hypotension and/or renal dysfunction favor invasive monitoring, there is no direct comparison of the efficacy of nesiritide versus nitroprusside. In favor of nesiritide, there is no need for dose adjustment for renal or hepatic dysfunction, no adverse coronary steal phenomenon(33) exacerbating ischemia, as there is with nitroprusside, nor is it necessary to monitor toxic metabolites such as thiocyanate, as with nitroprusside. In favor of nitroprusside is the established benefit of improved hemodynamics that can be sustained chronically (at 9- month follow-up) with appropriate adjustment of oral vasodilators and diuretics.(31) For patients who do not respond adequately to nesiritide, invasive monitoring with the addition of other intravenous therapies including nitroprusside and/or inotropes may be indicated.
Inotropic Support vs. Vasodilators for CHF
Intravenous inotropes have been utilized for the stabilization of decompensated heart failure since the 1970s. In a meta-analysis of the use of intravenous dobutamine and milrinone for the management of decompensated heart failure, there is no survival benefit, but rather a trend towards adverse outcomes.(34) Dobutamine, examined as the placebo/control arm of the Flolan International Randomized Survival Trial (FIRST)(35) and the control arm of the Levosimendan in Severe Low output Heart Failure study (LIDO),(35) and milrinone, in the OPTIME-CHF trial,(36) when used for the in-patient management of decompensated heart failure, were associated with increased mortality without improvement in rehospitalization. Trials of outpatient oral inotropes, in the early to mid 1990s, demonstrated similarly discouraging results.(37)
Some of the adverse outcomes associated with inotropes have been attributed to their tendency to induce tachyarrhythmias, including atrial fibrillation and ventricular tachycardia. Treatment necessary to control these accompanying arrhythmias may offset some of the hemodynamic advantages accrued from inotropic therapy. Nesiritide, unlike dobutamine and milrinone, is neither instrinsically chronotropic nor proarrhythmic. The Prospective Randomized Evaluation of Cardiac Ectopy with Dobutamine or Natrecor Therapy (PRECEDENT) study(38) demonstrated that nesiritide has a favorable safety profile compared to dobutamine and reduces or has a neutral effect on ventricular ectopy. The differences in hemodynamic properties of the intravenous agents commonly used in heart failure are summarized in Table 2.
Table 2. Properties of Intravenous Medications Used in the Treatment of Congestive Heart Failure
| Nesiritide | Dobutamine | Milrinone | Nitroprusside | |
| Inotropic | - | ++ | +++ | - |
| Increases cardiac index | + | +++ | +++ | ++ |
| Chronotropic | - | +++ | ++ | +/- (may cause reflex tachycardia) |
| Diruetic | + | - | - | - |
| Arrhythmogenic | - | + | + | - |
| Increase Myocardial Oxygen Consumption | - | +++ | +++ | +/- (may cause coronary steal) |
| Systemic vasodilator | +++ | +/- | + | +++ |
| Pulmonary vasodilator | ++ | - | ++ | + |
| Invasive monitoring | - | + | + | +++ |
| Renal dose adjustment | - | - | - | +++ |
| Toxic metabolities | - | - | - | + |
In addition to their detrimental chronotropic and proarrhythmic effects, inotropes increase myocardial oxygen consumption which can exacerbate cardiac ischemia in the setting of underlying coronary artery disease.
While inotropes have not been demonstrated to improve outcome, nesiritide holds promise for improving short-term mortality and reducing hospital length of stay and overall health care costs. In an open label study of 305 patients in 46 centers, nesiritide was compared to standard care (which included at least one of the following vasoactive medications: dobutamine, milrinone, nitroglycerin, nitroprusside).(39) A post-hoc analysis suggested that although overall hospital stay was not different between the two groups, the percentage of nesiritide-treated patients readmitted with heart failure within 21 days was lower. All cause readmissions were reduced from 20 to 10 percent. Furthermore, the nesiritide group required shorter treatment courses with IV vasoactive therapy and fewer additional parenteral agents. Six-month mortality rates were lower for the group receiving nesiritide.
Although the study size and absolute number of event rates were small, this study suggests that nesiritide may be more effective than dobutamine and have sustained long-term benefits.(39) Other studies have suggested that early administration of nesiritide compared with standard diuretic therapy is associated with shorter lengths of stay with comparable total volumes of diuresis.(40) The complete role of nesiritide in acutely decompensated heart failure remains to be defined, but early data show it to be a promising new agent in the treatment of heart failure.
Pulmonary Congestion with Salt and Water Retention
Nesiritide is indicated for patients with decompensated heart failure who demonstrate evidence of pulmonary congestion with salt and water retention. Absent frank cardiogenic shock, often defined as a systolic blood pressure less than 90 mm Hg, nesiritide is effective in facilitating diuresis, particularly in the presence of renal dysfunction. In the case illustrated earlier, nesiritide promoted efficient diuresis with minimal diuretic requirement in a patient who had been proven to be otherwise refractory.
Particularly in patients with atrial arrhythmias and/or frequent ventricular ectopy, nesiritide may be a safer treatment for acute decompensated heart failure. Unlike nitroprusside, nesiritide is not contraindicated in patients with renal dysfunction and is not associated with the accumulation of toxic metabolites. Baseline medications including beta-blockers, oral vasodilators and diuretics can be continued or adjusted as needed. As diuretic requirements have generally been shown to be decreased to achieve an equivalent level in volume reduction, a reduction in diuretic dose should be considered to avoid hypotension due be excessive hypovolemia. In our experience, patients who fail to respond adequately to empiric nesiritide within 12 to 24 hours often require invasive monitoring with the addition of inotropic agents and/or nitroprusside for hemodynamic optimization.
Future Applications of Nesiritide
While nesiritide received FDA approval following the VMAC trial, uncertainties remain about the long-term benefits of parenteral nesiritide use. Though several studies have demonstrated acute hemodynamic benefits, no large trials examining long-term outcomes have thus far been reported. Not dissimilar to the early reports of dobutamine, milrinone and epoprostenol(41) (Flolan), acute hemodynamic improvements do not necessarily translate to long-term benefit and may even be detrimental to survival.
The role of nesiritide continues to evolve. Whereas the preponderance of evidence currently does not support the use of continous and/or intermittent inotropes for the management of outpatient heart failure, recent reports have suggested a role for nesiritide. The Follow Up Serial Infusions of Nesiritide (FUSION) study was an open label pilot study to examine the safety and tolerability of a weekly single infusion of nesiritide over 12 weeks for patients with chronically decompensated heart failure compared with standard therapy including inotropes.(42) In patients considered high risk for rehospitalization, the study noted a trend toward improved functional status, fewer adverse events including rehospitalization, mortality and worsening renal function.
Although not powered to examine these clinical endpoints, these data favor a role of nesiritide in the outpatient management of chronic heart failure. Limited infusions of nesiritide may also play a role by decreasing the need for hospital admission in patients that could be treated for a 6 to 8 hour period in observation units of emergency departments.(40)
The potential roles of nesiritide in cardiovascular medicine continue to be expanded. The majority of patients enrolled in trials of nesiritide have had significant left ventricular dysfunction, although results from the VMAC trial suggest that the lusitropic effects of BNP may have significant benefit in the management of heart failure with preserved systolic function. The anti-proliferative, anti-fibrotic effects of the natriuretic peptides are also being explored in the prevention of adverse remodeling following acute myocardial infarction.(43) Further studies of nesiritide in the treatment of diastolic dysfunction and in the post-infarction remodeling are warranted. While small studies have demonstrated relative efficacy and safety of acute administration of nesiritide, larger randomized trials are needed to demonstrate both long-term safety and benefit. Furthermore, although early trials and economic modeling(44) would suggest that the reduction in length of stay, the decreased need for intensive care nursing and reduced rate of readmission with nesiritide would result in overall cost-effectiveness for a more expensive agent, the economic implications are as yet not fully determined.
Summary
BNP and NT-proBNP have been demonstrated to be useful biomarkers of heart failure that enhances diagnostic accuracy and may ultimately play an important role in the optimization of patient care. Elevated values are associated with a trend towards adverse outcomes. A reduction in BNP and NT-proBNP levels in response to therapy correlates with clinical improvement.
As BNP is a part of the endogenous counter-regulatory response to cardiac failure, recombinant BNP has been applied therapeutically to the management of acute decompensated heart failure. The preponderance of the evidence suggests that nesiritide is safe and well-tolerated with a superior safety profile to traditional inotropic agents. Its exact role in the management of heart failure continues to be expanded and offers promise as a novel agent in the management of both acute and chronic heart failure that may result in symptomatic benefits to the patient and economic benefits to the healthcare system.