Course Authors
Andrew D. Krystal, M.D., M.S.
Release Date: 07/08/2007
Upon completion of this Cyberounds®, you should be able to:
Describe new developments in research that have implications for changing the clinical management of insomnia
Discuss strategies for optimal clinical management of insomnia
Discuss the therapies that are available for the clinical management of insomnia and how to decide which to use in a given patient.
Dr. Krystal will discuss the unapproved use of antipsychotics, antidepressants and antihistamines for the treatment of insomnia.
Nearly everyone has difficulty sleeping at some point in their lives. A clinical diagnosis of insomnia is reserved for the subset of individuals experiencing a persistent problem falling asleep, staying asleep or achieving adequate restoration from sleep that results in impairment in their ability to function or their quality of life.(1) The percentage of the population meeting these criteria is substantial. It is estimated that 10-20% of the general population and 50% of patients seen in primary care suffer from clinically significant insomnia.(2),(3),(4),(5)
Despite the prevalence of this disorder, there is evidence that many affected individuals do not receive treatment and that much of the treatment administered is not empirically based.(6) Examination of the most common insomnia treatments administered in 2002 provides a vivid illustration of the degree to which practice is divorced from research (See Figure 1). Six of the ten most frequently administered treatments had not been studied in insomnia patients and the most frequently administered treatment had been studied once and failed to demonstrate efficacy.(7)

Figure 1. Controlled Studies in Insomnia Patients vs. Prescriptions in 2002.
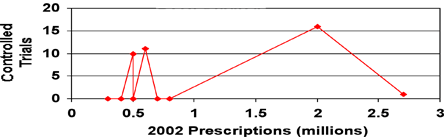
Many frequently prescribed agents have hardely been studied.
Figure 1 illustrates that one treatment that was prescribed 2.75 million times in 2002 was studied once; between 0.5 and 1 million prescriptions of two agents were written without any studies being carried out; between 0.25 and 0.5 million prescriptions were written for three other agents without a single study supporting their use in this population.

For many years we have lacked research studies focused on the sorts of issues facing clinical practitioners who treat insomnia patients. Such issues include the efficacy and safety of long-term treatment, and whether and how to treat insomnia occurring co-morbidly with medical and psychiatric conditions. Several recent studies, however, have begun to address these key issues in clinical practice. When viewed in combination with placebo-controlled studies that provide data on the safety and efficacy of an expanding set of insomnia treatments, these studies represent an emerging empirical basis to support the clinical practice of insomnia.
In this Cyberounds®, we review this evidence base and discuss strategies for its effective application in clinical practice. The discussion will focus on the following issues which highlight the changing point of view of the treatment of insomnia:
- Chronic insomnia is associated with significant impairment in function
- There is a need to treat insomnia when it is co-morbid with medical and psychiatric conditions.
- There are non-pharmacologic therapies that have been demonstrated to be efficacious in controlled trials.
- The long-term pharmacologic treatment of insomnia can be safe and effective and is not inevitably accompanied by dependence.
- The treatment of co-morbid insomnia not only improves insomnia but can improve the therapeutic response of the associated condition.
Who Should Receive Treatment for Insomnia?
The Importance of Impairment
Many people are significantly impaired by insomnia and could benefit from treatment.(4),(8),(9) Studies comparing chronic insomnia patients and normal sleepers suggest that affected individuals may experience daytime sleepiness, fatigue, cognitive impairment, symptoms of depression, symptoms of anxiety, a sense of impaired health or experience limitations in social and occupational function.(9)
While this is not surprising given that impairment is required in order to make a diagnosis, establishing that significant impairment often accompanies sleep disturbance is of great clinical relevance. For a clinician, the degree of associated impairment is probably the most important piece of information to obtain in order to decide whether treatment for insomnia is indicated. Analysis of risks and benefits dictates that the greater the associated impairment, the greater the need to institute treatment.
The Need to Treat Co-Morbid Insomnia
It is estimated that 80% of chronic insomnia occurs as a co-morbid condition with other medical or psychiatric disorders.(10) Persistent insomnia has long been viewed as a symptom of these associated disorders, thereby obviating the need for insomnia-specific treatment(11),(12) Long-standing clinical guidelines recommend that treatment be targeted to the underlying medical or psychiatric disorder, since insomnia, as a symptom of that disorder, should remit along with the other symptoms of the underlying condition.(12)
It is now clear that this point of view is problematic and inconsistent with the available research which suggests the need to treat insomnia when it occurs with medical and psychiatric disorders. One obvious problem with the symptom point of view is that it provides no guidance as to how to manage the 20% of insomnia patients who have primary insomnia without an associated, underlying treatable condition.(10) Further, several recent studies suggest that instituting effective treatment of supposed underlying medical and psychiatric conditions frequently does not resolve the associated insomnia.(13),(14),(15)
Recent data indicate that insomnia appears to be an independent predictor of adverse outcomes and future episodes of medical and psychiatric disorders.(10),(16),(17),(18),(19),(20) Based on these considerations, a 2005 NIH State-of-the-Science Conference panel proposed changing the commonly used term "secondary insomnia" to "co-morbid insomnia" and expressed the need to consider treatment of insomnia occurring in this setting.(2) Most compelling is new data (below) which provides evidence that treatment of co-morbid insomnia may not only improve insomnia but can also improve the outcome of the associated condition.(14),(15)
Insomnia Treatment Options
Choosing Among the Available Treatments
There are several pharmacologic and non-pharmacologic treatments available for patients with insomnia. Deciding which intervention is most appropriate for a particular patient should involve a review of the expected risks and benefits of each treatment options and is necessarily based on data from placebo-controlled trials. Though there are now placebo-controlled data available for many insomnia therapies, the absence of trials for some of the most commonly administered insomnia therapies is a severe obstacle to the judicious administration of these interventions in clinical practice.
It is not only necessary to have controlled data on an agent under consideration for insomnia patients, but data are also needed for the specific sub-population of insomnia patients that includes the patient being treated. This is particularly important in the treatment of children or the elderly where the risks associated with treatments may be greater or of a different nature from the treatment of adults aged 21-65 who have been the subjects in the vast majority of insomnia therapeutic trials. Of particular concern is that there has yet to be a single published study of a controlled-trial of the pharmacologic treatment of insomnia in children. Clinicans treating children are left with no empirical data on which to base their treatment decisions.
Other important sub-groups are those with medical and psychiatric co-morbidities such as major depression, anxiety disorders, chronic pain, etc. The likelihood of response and vulnerability to adverse effects of treatment may vary among those with different co-morbidities. While controlled data in these groups have been lacking, recent studies have been carried out on the treatment of insomnia occurring co-morbid with major depressive disorder, generalized anxiety disorder, rheumatoid arthritis, and fibromyalgia (See Table 6).
Efficacy has been demonstrated with both pharmacologic and non-pharmacologic therapies. Of particular note, in a large study of the treatment of insomnia occurring along with major depressive disorder (MDD), subjects treated with fluoxetine for their depression were randomized to adjunctive insomnia therapy with eszopiclone 3 mg or placebo.(14),(15) Those randomized to eszopiclone not only slept better but they had a more rapid and greater antidepressant response, which included improvement in features of depression other than sleep difficulties.
In the following two sections we present a summary of the literature of controlled-trials of insomnia therapies. This summary represents the available research base that can be used in making treatment decisions in clinical practice.
Non-Pharmacologic Therapies
A recent meta-analysis identified five interventions with empirical evidence of efficacy in the treatment of insomnia: stimulus control therapy, relaxation therapy, paradoxical intention, sleep restriction therapy and cognitive therapy.(21) Along with sleep hygiene therapy, probably the most common non-medication treatment for insomnia, these components are often administered in combination and several such combinations have been demonstrated to have efficacy in controlled trials.(22)
Sleep hygiene seeks to address behaviors and environmental factors that may contribute to disturbed sleep (see Table 1).(23)

Table 1. Sleep Hygiene Therapy.
| Caffeine | Limit use. Eliminate evening/night intake |
| Alcohol | Limit use. Eliminate evening/night intake |
| Nicotine | Eliminate smoking at night |
| Exercise | Eliminate exercising close to bedtime |
| Food/Liquid intake | Eliminate excessive intake close to bedtime but encourage eating enough not to promote waking up due to hunger |
| Light | Maintain a dark bedroom at night |
| Noise | Eliminate noise in the sleep environment |
| Temperature | Maintain comfortable temperature in bedroom |

Relaxation therapy is a series of interventions aimed at decreasing the level of arousal (see Table 2).

Table 2. Relaxation Therapies.
| Table Headline |
|---|
|

Paradoxical intention is based on the idea that, for some individuals, trying to stay awake actually decreases the anxiety and effort associated with falling asleep.(24) This therapy consists of instructing the patient to stay in bed and to try to stay awake as long as possible. Both relaxation therapy and paradoxical intention are primarily administered to address difficulties falling asleep.
Stimulus control therapy was developed in order to undo the conditioning that can develop from having many repeated nights marked by difficulty sleeping, frustration about sleep and anxiety in the same setting.(21) This treatment involves minimizing the likelihood of being awake, frustrated or anxious in the sleeping environment (see Table 3).

Table 3. Stimulus Control Therapy.
| Table Headline |
|---|
|

Sleep restriction therapy is an effective treatment, though poorly named.(21) If successful, the amount of sleep achieved remains unchanged; however, the time in bed is reduced and, as a result, sleep is better consolidated. This is achieved by choosing a consistent bed time and wake up time that correspond to the average amount of sleep that the patient is actually getting. This process is greatly aided by obtaining a diary of sleep/wake function over the preceding 1-2 weeks. Other important elements of this therapy are eliminating naps and instructing the patient never to go to bed before the prescribed bedtime; however, in order not to contribute to adverse conditioning, it is also important to instruct the patient not to go to bed after this bedtime until they are sleepy. If the prescribed time in bed is too short, the patient will become excessively sleepy and the prescribed time in bed should be lengthened by 15-30 minutes. If, on the other hand, the insomnia continues, the time in bed should be further curtailed by 15-30 minutes.
Cognitive therapy is targeted to dysfunctional beliefs and thought processes that tend to disturb sleep. Examples include the assumption that not getting 8 hours of sleep on any given night will have a disastrous effect on function the following day and the belief that waking up in the middle of the night is always a sign of sleep disturbance.
Risk-Benefit Considerations for Non-Pharmacologic Therapies
While it is often believed that non-pharmacologic therapies are without adverse effects, there are some potential negative consequences that should be considered in making treatment decisions. With sleep restriction therapy, patients may experience excessive daytime sleepiness during the first several weeks or in instances where the prescribed time in bed is too short.(25) ) Other considerations are the monetary cost of therapy and the delay of up to several weeks before the therapeutic effect is fully realized. Further, if treatment is unsuccessful, the attempt to institute therapy will have increased the time before the insomnia is resolved. On the other hand, when these therapies are effective, the benefit is typically long-lasting.(21)
In assessing the likely benefit, the most important considerations are the willingness and ability to implement the therapy as well as the presence of behavioral targets.(26) Implementing behavioral changes to improve sleep can be challenging just like changing behaviors in order to lose weight can be very difficult for some individuals. Without a high level of motivation, non-pharmacologic therapies are doomed to fail. Further, some subgroups of insomnia patients are unable to institute some or all of the prescribed behavioral interventions. This includes those with medical illnesses that require them to be bed-bound and those with disorders such as severe major depression, dementia, mental retardation and schizophrenia.
Lastly, only a subset of insomnia patients actually have maladaptive behaviors and cognitions that play a role in perpetuating their insomnia. It is only in this subset of patients that cognitive-behavioral interventions are likely to be effective treatments for insomnia. When evaluating the suitability of non-pharmacologic therapies in a patient with insomnia, it is important to determine the degree to which there are contributing maladaptive behaviors and cognitions that can be targeted with treatment.
Pharmacologic Therapies
Agents from a number of medication classes are commonly used to treat insomnia. These include: benzodiazepines, non-benzodiazepines, melatonin receptor agonists, antidepressants, antipsychotics and antihistamines.(2) Of these, only the benzodiazepines, non-benzodiazepines and melatonin-receptor agonists are approved by the U.S. Food and Drug Administration (FDA) for the treatment of insomnia. The benzodiazepines represent a class of agents with related chemical structure that all act via binding sites on the gamma amino butyric acid (GABA) receptor complex. The non-benzodiazepines are structurally unrelated to the benzodiazepines, or each other, but have in common that they also act via the same GABA binding sites as the benzodiazepines.
There are a variety of types of GABA receptor complexes distributed throughout the brain and the non-benzodiazepines appear, at least in animal models, to have relative specificity for the subtype of GABA receptor complexes that modulate sleep, resulting in greater specificity of effect and, therefore, the potential of diminishing some types of diverse effects.
Recently, a selective agonist at type-1 and type-2 melatonin receptors, ramelteon, also became available for treatment of sleep onset insomnia.(27) The antidepressants used to treat insomnia are a heterogenous group comprised of tricyclic antidepressants (including amitriptyline, doxepin and tri-imipramine), serotonin receptor antagonists (mirtazapine and nefazodone) and trazodone, which is a serotonin receptor antagonist and inhibitor of serotonin reuptake. These agents have varying degrees of anti-histaminergic effects which, along with adrenergic and serotoninergic antagonism, are thought to mediate their sedating effects.(15) The antipsychotics enhance sleep via these same mechanisms and those most commonly used to treat insomnia include quetiapine and olanzapine.(28) Anti-histamines are the most commonly used over the counter sleep aides.(2)
In deciding which of these many agents to administer to a given patient with insomnia, it is necessary to determine whether an agent has demonstrated efficacy for the type of problem that the patient is experiencing and weigh this evidence against the expected adverse effects profile. As discussed above, the minimal empirical support for the efficacy of any of the agents other than the FDA approved agents (benzodiazepines, melatonin receptor agonists and non-benzodiazepines) impedes their effective use. Table 5 lists the agents and dosages that have been demonstrated to have efficacy in treating insomnia.

Table 5. Studies Demonstrating Insomnia Efficacy.
| Agent | Sleep Onset | Sleep Maintenance | Longest Controlled Trial | Older Adults |
|---|---|---|---|---|
| Benzodiazepines | ||||
| Triazolam 0.25 mg | + | 4 Weeks | + | |
| Triazolam 0.5 mg | + | + | 2 Weeks | + |
| Flurazepam 30 mg | + | + | 4 Weeks | + (15-30 mg) |
| Estazolam 1/2 mg | + | + | 2 Weeks | |
| Quazepam 30 mg | + | + | 2 Weeks | |
| Temazepam 30 mg | + | + | 8 Weeks | + (7.5-30 mg) |
| Non-Benzodiazepines | ||||
| Zolpidem 10 mg | + | 5 Weeks* | + (5 mg) | |
| Zolpidem CR 12.5 mg | + | + | 3 Weeks | |
| Zaleplon 10 mg | + | 5 Weeks | + (5-10 mg) | |
| Eszopiclone 2/3 mg | + | + | 6 Months | + (2 mg) |
| Melatonin Receptor Agonists | ||||
| Ramelteon 4-32 mg | + | 5 Weeks | + | |
| Antidepressants | ||||
| Doxepin 25-50 mg | + | + | 4 Weeks | |
* There is also a 3-month placebo-controlled study of intermittent dosing.(27),(29),(30),(31),(32),(33),(34),(35),(36),(37),(38),(39)

This table also indicates whether benefits have been established for onset problems and/or sleep maintenance problems, the longest duration of demonstrated efficacy in a placebo-controlled trial and whether there is evidence of efficacy in older adults. Table 6 lists therapies (both pharmacologic and non-pharmacologic) demonstrated to have efficacy in co-morbid insomnia treatment.
In weighing the likelihood of efficacy against the risks of adverse effects for an agent, it is best to consult the FDA approved label which provides an indication of the adverse effects rate for that agent vs. placebo. The potential impact of side effects is greatest in older adults The most common side effects of benzodiazepines and non-benzodiazepines are sedation, dizziness, amnesia and ataxia.(2) The antidepressants and antipsychotics can be associated with a variety of effects which depend on the agent and may include sedation, dizziness, orthostatic hypotension and anticholinergic effects.(2) The available over-the-counter sedating anti-histamines are most often associated with daytime sedation, dizziness, and anti-cholinergic side effects.(2)
Clinical Implementation: The Importance of Follow-up
Among the most important elements needed to effectively apply the available insomnia research in clinical practice is follow-up assessment. For non-pharmacologic therapies this is needed in order to work through the difficulties that are nearly universal when attempting to change behavior. Further, modifications of the prescribed behavioral regimen are often needed in order to achieve optimal benefit. The role of follow-up in pharmacotherapy is no less important. It is crucial to re-evaluate patients to decide if the prescribed medication is effective and to determine if there are side effects. Often adjustments in dosage are needed or a different agent prescribed. Any potential safety issues identified must be addressed.
Another crucial function of follow-up in pharmacotherapy is to decide when to stop therapy. We currently lack any means to determine how long an individual with insomnia is likely to continue to have sleep difficulties. Further, once pharmacotherapy is initiated, there is no way to determine when the insomnia has remitted. As a result, it is necessary to make a contract with the patient at the beginning of the course of pharmacotherapy that at some point in the future (typically every 2-3 months) a trial of medication discontinuation will be carried out. This should be done with a very gradual taper.
After the medication has been discontinued, an analysis of the relative risks and benefits of continuing the medication, discontinuing pharmacotherapy or switching to another agent can be carried out. In those who were overall doing better with their medication than without, reason would dictate continuing treatment with this medication. As such, long-term pharmacotherapy cannot be determined a priori but occurs only through a series of follow-up risk-benefit assessments that repeatedly favor the ongoing use of medication.
As can be seen in Table 5, the sustained safety and efficacy of a few agents have been established in controlled-trials for periods of over one month and only one agent has been evaluated as long as six months and there was no evidence of loss of benefit or increase in risks over time.(31) However, these studies open the door to other studies as they establish that dependence phenomena (tolerance and withdrawal) are not inevitably associated with longer-term insomnia pharmacotherapy and that treatment, at least as long as six months, can be safe and effective with some agents in those individuals with insomnia where extended treatment is needed.
Conclusion
The field of insomnia treatment research is evolving rapidly. While it has historically been inadequate to serve as a basis for effective clinical practice, this situation is changing. Developments of particular importance for impacting clinical practice include: the evidence of the functional impairment associated with insomnia, data supporting the need to treat co-morbid insomnia and the potential benefit of doing so, studies that established the efficacy of non-pharmacologic therapies, and the research demonstrating that long-term pharmacotherapy is not inevitably associated with dependence phenomena. Effective implementation of this research base also presents challenges; however, ongoing work in both further expanding the research base and on its clinical application promises to lead to a substantive advance in optimizing the clinical treatment of insomnia.