Course Authors
Ian J. Griffin, M.D.
Release Date: 09/15/2007
Upon completion of this Cyberounds®, you should be able to:
Describe the presentation, pathophysiology and treatment of acrodermatitis enteropathica
Discuss the effect of zinc supplementation on childhood morbidity and mortality in developing countries
Describe the cellular transport of zinc and the different roles of ZIP and ZnT transporters
Discuss the pathophysiology of breast-feeding associated zinc deficiency and be able to differentiate it from acrodermatitis enteropathica.
This work is a publication of the U.S. Department of Agriculture (USDA)/Agricultural Research Service (ARS) Children's Nutrition Research Center, Department of Pediatrics, Baylor College of Medicine, and Texas Children's Hospital, Houston, Texas. This project has been funded in part with federal funds from the USDA/ARS under Cooperative Agreement number 58-6250-6-001, the NIH, NCRR General Clinical Research for Children Grant number RR00188. Contents of this publication do not necessarily reflect the views or policies of the USDA, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. government.
Zinc is a component of over 250 enzymes and is essential for human health and development.(1) Zinc atoms can play either a structural role within proteins or a catalytic one.(2) Structural zinc atoms are bound to four cysteine or histidine residues creating a tetradentate site.(2),(3) Catalytic zinc atoms form a tridentate active site that is bound to three cysteine, histidine, glutamate or aspartamate residues, with the fourth coordination site occupied by an activated water molecule.(2) Zinc metalloproteins are important for many cellular functions including DNA and RNA replication, gene transcription and apoptosis.(4) Given the increasing interest in zinc biochemical functions, it is surprising that recognition that zinc was essential in humans is a relatively recent event.(5)
Historical Description of Zinc Deficiency
In 1961 Prasad et al. described a group of 11 Iranian men with a syndrome of iron deficiency, dwarfism, geophagia and hypogonadism.(6) Symptoms responded well to a mixed diet but the cause of the syndrome was unclear, as iron deficiency alone could not explain all the features seen.(6) A few years later, a similar set of clinical features was noted in a group of Egyptian men.(7) As with the Iranian subjects, their diet was very limited, included very little animal protein and there was pronounced geophagia (pica).
The Egyptian subjects were studied in-depth and found to have abnormally low zinc concentration in the plasma and red blood cells, an increased turnover of radio-labeled zinc from the plasma, a reduced size of the exchangeable zinc pool and decreased urinary and fecal zinc excretion. Their growth increased dramatically with zinc supplementation. Serum alkaline phosphatase (a zinc-containing enzyme) rose significantly with treatment,(7) as had happened previously with the Iranian subjects.(6) Taken together, this data provide compelling evidence that both groups of subjects had zinc deficiency in addition to other nutrient deficiencies.
Acrodermatitis enteropathica (see below) is a congenital disorder of zinc malabsorption(8) that was first described in 1936,(9) although it was only identified as a disorder of zinc metabolism in 1976,(10) more than a decade after Prasad's description of zinc deficiency in Iran(6) and Egypt.(7)
Review of Zinc Homeostasis
Zinc is absorbed throughout the gut but mostly in the small intestine. The highest rates of zinc absorption occur in the jejunum(1),(11) but the total amount of zinc absorbed may be highest in the duodenum, as intraluminal zinc content is greater in the duodenum than in the jejunum.(12) Most absorption is active and saturable, although paracellular absorption can occur at very high zinc intakes. Zinc is excreted in the feces (largely from pancreatic and biliary secretions) and in the urine.(1),(3)
Fractional zinc absorption decreases as the zinc load increases. In adults given supplementary zinc as an aqueous solution, fractional zinc absorption rapidly decreases as dose increases;(13) at intakes above 20 mg, relatively little additional zinc is absorbed.(13) However, estimates of zinc absorption from single doses are likely to overestimate zinc absorption when supplements are given more regularly. Tran et al., for example, have shown that fractional zinc absorption from a 20 mg test dose is reduced by 40% if a 20 mg dose was given the preceding day.(13)
Zinc homeostasis is a balance between zinc absorption and zinc losses in the urine and feces, although changes in urinary zinc excretion only occur very late in zinc deficiency.(4) It has been suggested that changes in absorption provide "coarse" control of zinc homeostasis, while changes in excretion may provide "fine" control.(4) Zinc depletion does lead to dramatic decreases in endogenous losses of zinc in to the feces(1) but its effect on zinc absorption is less clear.
Lee et al. carried out a long-term study(14) where subjects were placed on a low zinc diet for 6 months. Zinc absorption initially increased on the zinc deficient diet after 2m and 4m but by 6m the increase was no longer statistically significant from baseline. Endogenous fecal zinc excretion, in contrast, continued to decline during the entire 6-month study period. However, the zinc isotope was given with a lower zinc load in the test meal during the depletion period, so it remains unclear whether the increase in fractional zinc absorption was a response to worsening zinc status or simply from lower zinc load. Similarly, data from Wada(15) show a significant increase in zinc absorption 7-15d after the start of a 5.5 mg/d zinc diet compared to subjects consuming 16.5 mg/d zinc. However, this seems more likely to have been a response to the lower zinc load in the meal rather than an effect of developing zinc deficiency. This issue was clarified in a short-term study by Istfan et al.(16) After 24d on a zinc deficient diet, zinc absorption was significantly greater compared to a group fed a zinc adequate diet, even though both groups had received a similar zinc load in the test meal.(16) An alternative study considered the effect of increasing zinc intake.(17) As zinc intake increased so did endogenous fecal zinc excretion. However, there was a delay of several days before zinc absorption responded to the increased zinc intake and the absolute zinc absorption briefly rose following increased zinc intake before returning to baseline.(17)
A very different study design was utilized by Sian et al. (18) Instead of using experimentally controlled zinc depletion, these investigators measured zinc absorption in two groups of Chinese women whose usual zinc intakes were 5.2 mg/d and 8.1 mg/d. Zinc absorption did not differ between the two groups but endogenous fecal zinc excretion was significantly lower in the group with the lower zinc intake(18) suggesting that changes in endogenous zinc excretion in the gut are the principal source of homeostasis under normal conditions.
In summary, endogenous fecal zinc excretion is a major site of zinc homeostasis. Fractional zinc absorption falls as zinc intake increases, but it remains unclear how much changes in fractional zinc absorption contribute to zinc homeostasis, particularly in the long term.
Zinc Transporters
Irrespective of whether changes in zinc absorption or endogenous fecal zinc excretion are the primary site for zinc homeostasis, it seems likely that these processes must utilize specific transporters in order to achieve their function and to allow regulation of absorption and excretion in response to changes in zinc status. The first mammalian zinc transporter, ZnT1, was identified in 1995.(19) Since then more than 20 have been described.(20)
Zinc transporters fall into one of two large families. The ZIP (SLC39A) family is responsible for zinc influx to cells and, therefore, increase intracellular zinc concentration.(20) The ZnT transporters (solute-linked carrier 30 family, SLC30A) are responsible for zinc efflux from cells which thus reduces intracellular zinc concentrations.(20)
The ZIP family (Zrt- and Irt-like proteins) is named for its homology to the yeast (Saccharomyces cerevisae) IRT1 (iron regulated transporter) and the Arabidopis thaliana proteins ZRT1 and ZRT2 (zinc regulated transporter).(21),(22) Most family members are predicted to have 8 transmembrane domains, extracellular N- and C-terminal tails (23) and a variable intracellular histidine-rich domain that may be important for metal binding.(21),(22) The first family member to be identified was originally described as an iron transport protein but it is now known to be able to transport zinc and manganese as well.(21) Subsequently, three related Arabidopis proteins were identified that appear to be able to transport zinc but not iron.(21)
All known SLC39A family members transport metals into the cytoplasm either from intracellular compartments or from outside the cell across the plasma membrane.(22) At least 14 human members of the SLC39A family have been described and many of them (ZIP1 through ZIP8 and ZIP14) appear to transport zinc.(20) The mechanism of action is, however, poorly understood(20), although it is known to be energy independent.(22) Zinc transport by ZIP1 and ZIP2 is not dependent on ATP or on Na+ or K+ concentration gradients.(20),(22)
The expression of many ZIP proteins is affected by zinc concentration.(24),(25) ZIP1 is widely expressed and is up-regulated by zinc deficiency.(24) ZIP2 is also zinc-regulated as shown in mononuclear cells.(24) ZIP3 is highly expressed in bone marrow and spleen,(24) as well as in mammary epithelium where it is up-regulated by estrogen.(20) ZIP14 is up-regulated in the liver by inflammation and appears to mediate the fall in plasma zinc that is seen as part of the acute phase reaction.(26) This effect is partly mediated by IL-6, which increases expression of ZIP14 mRNA.(26) The fall in plasma zinc that occurs in inflammation is blunted in IL-6 knockout mice.(26)
Much interest has centered on ZIP4. This is expressed most highly in tissues that are important in zinc homeostasis -- the small intestine, stomach, colon and kidney.(22),(24) Mutations in this gene are now known to cause the hereditary disorder of zinc malabsorption and deficiency, acrodermatitis enteropathica (see below).(27),(28)
The ZnT proteins are also known as cation diffusion facilitators (CDF)(23) and have been more extensively studied that the ZIP proteins. They show significant homology, with most members having 6 transmembrane domains,(20) except for ZnT5 which has 12 or 14.(20),(23) They have cytoplasmic C- and N-termini and a large histidine-rich intracellular loop.(23) Four of the transmembrane domains form a channel and the transporters may function as homo- or hetero-dimers.(20) They may also take part in protein-protein interactions and be involved in the insertion of zinc into the zinc metalloprotein, alkaline phosphatase.(20)
ZnT1 was first identified as conferring protection against high zinc concentrations in cell culture.(24) It is widely expressed, especially in tissues important in the maintenance of zinc homeostasis such as the small intestine, renal tubules and the placenta.(24) ZnT1 is mainly localized to the basolateral membrane of the enterocyte where it acts to transport zinc out of the cell to the circulation.(23) In the kidney, ZnT1 is expressed in the loop of Henle and may act to re-absorb zinc from the urinary filtrate.(24) Both ZnT1 and ZnT2 are expressed in the pancreas where they are down-regulated in zinc deficiency.(20) This may be an adaptive mechanism which results in decreased endogenous zinc losses in pancreatic secretions. ZnT1 is probably the only member to be sited at the plasma membrane and so plays a major role in efflux of zinc from the cell.(20) However, it can also be localized intracellularly, for example in vesicles in the pancreatic acinar cells.(20)
ZnT2 also improves tolerance to high zinc levels.(24) It is less widely expressed than ZnT1 and is found in the prostate, small intestine and kidney.(24) It is predominantly found in intracellular vesicles.(20)
ZnT4 is found in the mammary gland, small intestine and the brain (24). In mice, it leads to the lethal milk (lm) phenotype.(24) In this disease, maternal zinc levels in breast milk are very low and pups die of zinc deficiency before weaning.(24)
ZnT5 is found in the pancreas (particularly the insulin secreting β-cell) where it co-localizes with secretory vesicles,(20) the ovaries, testis and prostate.(24)
The effect of zinc intake on the expression of some of the ZnT proteins has been studied in experimental animals, particularly the rat. Liuzzi et al.(29) examined expression of ZnT1, ZnT2 and ZnT4 in rats fed a basal diet, a zinc supplemented diet and a zinc deficient diet. Zinc status most affected ZnT4 expression, followed by ZnT1 and lastly by ZnT2. Zinc supplementation increased ZnT1, ZnT2 and ZnT4 expression in the kidney and small intestine. Zinc restriction had little effect on ZnT1 expression in the small intestine but did reduce expression of ZnT2 and ZnT4 in small intestine. Zinc restriction reduced the expression of ZnT2 but not ZnT1 and ZnT4 in the kidney. Both ZnT1 and ZnT2 are down-regulated in the pancreas in response to zinc deficiency.(20) Liver expression of ZnT1 and ZnT4 are unaffected by zinc supplementation or by zinc restriction.(29) Expression of ZnT1, ZnT2, but nor ZnT4, falls acutely after a large oral load of zinc.(29) This may partly explain the decrease in zinc absorption that occurs immediately following a large oral dose of zinc.(13)
The effect on zinc and vitamin deficiency on ZnT1, ZnT2 and ZnT4 expression in mammary glands has also been studied in rats.(30) ZnT1 expression is unaffected by zinc deficiency, vitamin A deficiency or combined Zn and vitamin A deficiency. ZnT1 protein levels are, however, reduced in all three deficiency conditions.(30) Neither zinc nor vitamin A deficiency affects ZnT2 expression or ZnT2 protein levels. Zinc deficiency significantly increases ZnT4 expression and protein content. Vitamin A deficiency had a smaller but still significant effect.(30) Surprisingly, combined vitamin A and zinc deficiency had no effect on mRNA or protein levels.(30)
The effect of zinc supplementation of expression of zinc transporters has been examined in human leukocytes.(31) Zinc supplementation leads to large increases in ZnT1 and ZnT7 expression and significant falls in ZIP3 and ZIP1 expression.(31) Significant changes in ZIP1 mRNA and ZIP3 mRNA were apparent within 2d of starting zinc supplementation and they returned to baseline within 4d of stopping supplements.
Physiological Role of Zinc Transporters
The preceding brief overview of zinc transporters serves to show how complex and interrelated the maintenance of zinc homeostasis is. It is also likely that there is much overlap of function between the different transporters of the same family.
A simplified schematic of zinc homeostasis has been suggested by Cousins (Figures 1A and 1B).(20)

Figure 1A. Basal (normal zinc) Conditions.
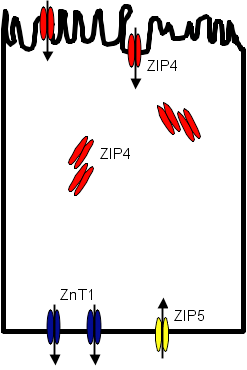
Figure 1B. Low Zinc Conditions.
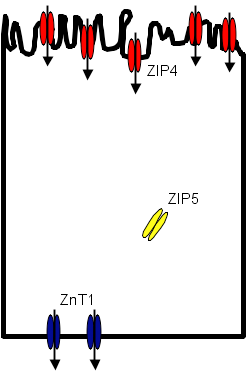

Under normal conditions ZIP4 is expressed in intracellular storage sites as well as at the apical surface of the enterocyte where it transports zinc into the cell.(20) ZnT1 is expressed at the basolateral surface where zinc effluxes to the circulation. Some zinc is taken up from the circulation back to the enterocyte at the basolateral surface through ZIP5, reducing the net efflux of zinc out of the cell. In response to zinc deficiency, more ZIP4 is synthesized and intracellular stores localize to the cell membrane, increasing the amount of receptor at the apical surface of the cell and increasing the amount of zinc taken up by the cell. At the same time, ZIP5 is re-localized from the basolateral membrane to intracellular sites, reducing the reverse transport of zinc into the enterocyte from the circulation. Efflux of zinc via ZnT1 is now not counteracted by influx through ZIP5 and the forward transfer of zinc from gut lumen to enterocyte to circulation increases.
In pancreatic acinar cells, ZIP5 allows entry of zinc into cells with exits into pancreatic secretions via ZnT1 and ZnT2 (mostly in transport vesicles). In response to zinc deficiency, ZIP5 is internalized and entry of zinc into the acinar cells falls. At the same time, ZnT1 and ZnT2 are down-regulated and the efflux of zinc into pancreatic secretions decreases. This model would serve to increase gastrointestinal absorption of zinc and reduce losses of zinc in pancreatic secretions at times of reduced zinc status.
Despite the large numbers of zinc transport proteins, few diseases seem to be directly caused by mutations in these transport proteins. We will, however, consider two such diseases -- acrodermatitis enteropathica and breast-feeding associated zinc deficiency.
Acrodermatitis Enteropathica
Acrodermatitis enteropathica (AE, OMIM 201100) is the prototypical disease of zinc deficiency in humans.(32) It is an autosomal recessive disorder of zinc malabsorption presenting with diarrhea, dermatitis and alopecia. (33) The skin rash can be pustular or hyperkeratotic and is most commonly found periorally and on extensor surfaces of the limbs. Its other features include ocular findings (conjunctivitis, keratopathy), irritability, apathy, depression, tremors, ataxia and photophobia, angular chelitis, paronychia, lactose intolerance, failure to thrive, delayed puberty and hypogonadism, and recurrent infections.(8),(32),(34) The incidence of AE is estimated to be 1:500,000 with no known gender or race preponderance.(33) Serum zinc is usually, but not always, low and serum lipids are abnormal.(8) A serum zinc less than 50 mcg/dL is usual but the clinical picture of AE that responds to zinc supplementation can be seen with normal serum zinc concentration.(35)
The disease was first described in 1936, although the cause was unknown and it was ultimately fatal.(9) It was serendipitously found that clinical features of AE were improved by treatment with diodoquin (an antimalarial),(34) although this treatment was associated with the development of an irreversible retinopathy.(33) In retrospect, diodoquin probably acted as a zinc ionophore (a lipid-soluble molecule that helps transport an ion across a membrane) and increased zinc absorption;(36) amphotericin may act in a similar fashion.(37)
In 1974 Moynahan demonstrated that AE was a zinc deficiency disease and could be treated with supplementary oral zinc.(38) AE is usually treated with 35-100 mg/d, or 3 mg/kg/d(8),(33) elemental zinc by mouth, although zinc requirements often fall after puberty.(8) High levels of zinc supplementation are required, consistent with a defect in zinc absorption.(39) Clinical improvement occurs rapidly once zinc supplementation is started and well before serum zinc becomes normal.(33) Zinc absorption is reduced in AE despite the zinc deficiency.(40) The defect is most apparent in children(40) and affects both the jejunum and duodenum.(10),(41),(42) Abnormal jejunal zinc transport was seen in biopsy samples from subjects with AE and investigators concluded that the "AE mutation affects both zinc binding to the cell surface and its translocation across the plasma membrane into the cell, possibly mediated through a defective anionic exchange mechanism."(43) Although the gut is central to the pathophysiology of AE, metabolism defects extend more widely, with zinc uptake from cultured fibroblasts for patients with AE being abnormal.(44)
The genetic basis of AE is now known. The abnormal gene loci was mapped to the 8q24.3 region, which was known to include a zinc transport protein, ZIP4.(22) In 2002, two groups independently identified mutations in the ZIP4 gene in unrelated subjects with AE(27),(28) confirming its role in the etiology of AE.
Breast-feeding Associated Zinc Deficiency
Acrodermatitis enteropathica usually presents shortly after infants are weaned. A somewhat similar disease has been described in breast-fed infants. There are several case reports of term and preterm infants presenting with clinical features of zinc deficiency which resolve rapidly once infants are weaned or modest dose zinc supplements started.(45),(46),(47) Cases respond rapidly to dietary diversification and do not recur if zinc supplements are discontinued.(48)
In many of these cases the zinc concentration in breast milk is very low and it was speculated that "inadequate breast milk zinc is thought to result from a defect in transfer of zinc from maternal serum to breast milk."(48) The condition also appeared to be hereditary. For example, Sharma described an extended family with 10 affected members -- all had symptomatic zinc deficiency appear before weaning while the infants were being breast-fed.(49) It differs from AE in that:
- The condition responds to weaning or the provision of dietary zinc sources
- The condition tends to occur before weaning and
- Zinc absorption is normal.(50)
In many ways, this disease is similar to lethal milk syndrome in mice, an autosomal recessive disease where maternal milk concentration is very low and pups develop zinc deficiency before weaning that is often fatal. In pups who survive the neonatal period (by cross-fostering the pups to normal mothers), they are subsequently normal until about 8m of age when they again develop zinc deficiency.(24) Although infants with breast-feeding associated zinc deficiency do not seem to develop later zinc deficiency, the parallels between the two diseases are obvious. The murine disease results from mutations in the ZnT4 protein.(24) However, in two unrelated cases of breast-feeding associated zinc deficiency, no mutation in ZnT4 was seen.(51) Subsequently, defects in another ZnT protein, ZnT2, were shown to be responsible for the condition. Chowanadisai described two unrelated cases that both had the same missence mutation in ZnT2, with replacement of a histidine residue by arginine.(52) Although the human and murine conditions have similar phenotypes, they are caused by mutations in two different, albeit related, proteins.
Zinc Supplementation of Healthy Infants
Breast-feeding associated zinc deficiency is extremely rare and few are likely ever to see a case. However, breast-fed infants grow more slowly than formula-fed infants, even in the U.S., and it has been speculated that this may in part be explained by differences in zinc intake.(53) In one small controlled trial, 4- to 10-month-old breast-fed infants were randomized to receive 1 mg/kg/d zinc or a placebo.(53) No differences in growth were observed and their appeared to be no benefit from zinc supplementation.(53) The same is not true in many developing countries were several studies have shown that zinc supplementation of high-risk infants has several benefits, including improved growth.(54),(55)
Zinc in Developing Countries
Although breast-fed U.S. infants may not require supplementary zinc, the same is not true in developing countries. Many(56)(57) but not all(58) studies have suggested that zinc supplementation of at-risk populations during the first year of growth has beneficial effects on growth.(54) The reason why not all populations benefit from zinc supplementation is incompletely known(55) but those most likely to benefit are older children with lower serum zinc concentrations and those who are stunted prior to intervention.(54)
Zinc has also received much attention for its possible effect on reducing childhood mortality and morbidity in developing countries. Although space does not permit a detailed review, this possibility has been discussed at length previously.(59),(60),(61),(62),(63)
Acute and chronic diarrhea led to 2.1 million deaths in children aged less than 5y in 2000,(60) and 1.6 million deaths in 2002.(64) The vast majority of these deaths occurred in developing countries and up to 88% were potentially preventable.(60) Zinc supplementation has clearly been shown to decrease the mortality and morbidity from acute and chronic diarrhea.(60),(65) One meta-analysis has estimated an odds ratio of diarrhea prevalence to be 0.75 in zinc supplemented children compared to controls.(61)
Prophylactic use of zinc in developing countries could reduce deaths among children aged less than 5y by between 350,000-450,000 annually (equal to 5% of all deaths in this age group).(60) Use of therapeutic zinc during episodes of diarrhea could prevent a further 5% of deaths (394,000), more than would be saved by effective neonatal resuscitation programs and almost as many as the appropriate use of antimalarial medications.(60) The WHO and UNICEF now recommend that the treatment of acute diarrhea include zinc supplementation (20 mg/d, or 10 mg/d if aged less than 6m) for 10-14d during the acute episode.(64)
Pneumonia causes almost 2.1 million deaths among children (age >5y) in developing countries of which 1.3 million (65%) are potentially preventable.(60) Again, there is compelling evidence that zinc treatment reduced the mortality of pneumonia among children in developing countries(61),(65) and may reduce the odds ratio for pneumonia by over 40%.(61)
Zinc and the Common Cold
Although it is far less of a healthy concern than pneumonia and diarrhea in developing countries, the possible role of zinc in reducing the severity and duration of the common cold has attracted much attention. Two main types of zinc therapy have been studied -- zinc lozenges taken by mouth or intranasally-administered zinc sprays.
Zinc lozenges have been most extensively studied. Early studies suggested that zinc gluconate lozenges could significantly reduce the duration of naturally occurring colds(66) and those induced experimentally.(67) Other studies suggested that zinc gluconate lozenges reduced the mean symptom duration from 7.6d to 4.4d(68) and that zinc acetate lozenges reduced the duration of some symptoms between 25 and 50%.(69) This degree of reduction is similar to that which people consider to be clinically significant and worthwhile.(70) However, other studies have been unable to replicate these findings.(71)(72),(73)
There has been a concern as to how well blinded subjects are in some of these studies.(74) Some studies have reported the incidence of side-effects (e.g., bad taste, nausea, etc.) to occur in 56-80% of subjects treated with zinc, compared to 30% or less of those treated with placebo.(66), (68) Not only does this raise questions about subjects becoming un-blinded by the metallic taste of zinc supplements, it makes it difficult to balance the advantages of a possible (but uncertain) diminution of viral symptoms with the disadvantages of the associated side-effects (albeit generally mild) of treatment.
In experimentally induced colds, some studies report more rapid improvement in symptoms and reduced amounts of nasal discharge following zinc treatment,(67) while others report no benefit and increased amounts of nasal discharge following zinc treatment.(71) Of note is the fact that the positive study only examined 12 subjects with experimentally induced colds.(67) A much larger study (N=273) compared symptoms after treatment with placebo, zinc gluconate and zinc acetate (using 2 different doses).(75) Zinc gluconate reduced disease duration from 3.5d to 2.5d but zinc acetate lozenges had no effect. Neither zinc acetate nor zinc gluconate lozenges had any effect on naturally occurring colds.
One study has examined the effect of an intranasal zinc spray on duration of common cold symptoms and found no beneficial effect.(76) It also seems prudent to avoid these preparations, as they may cause anosmia that is distinct from post-viral anosmia.(77)
In summary, zinc nasal sprays are best avoided and the usefulness of zinc lozenges is questionable, and they are not recommended by the American Academy of Family Physicians.(74) This possible role for zinc can only be clarified by much larger, well blinded, randomized controlled studies, and not by weaker designs such as retrospective chart reviews(78) or open label studies.(79)
Summary
There have been considerable recent advances in understanding the molecular physiology which underlies zinc metabolism and homeostasis. Much has been learned about the zinc transport proteins and two inherited human diseases known to be caused by mutations in these proteins. These diseases are very rare, however, and globally their importance is dwarfed by the emerging role of zinc supplementation to prevent mortality and morbidity in high-risk populations of children of developing countries. If properly applied, zinc supplementation and the therapeutic use of zinc (e.g., in acute diarrhea) has the potential to save the lives of millions of children annually. In healthy subjects in developed countries, the role of, and need for, zinc supplementation is far less certain.