Course Authors
Edward Saltzman, M.D.
Release Date: 12/10/2004
Upon completion of this Cyberounds®, you should be able to:
List foods which are the primary determinants of dietary vitamin K intake
Discuss potential interactions between vitamin K and vitamin E, and between vitamin K and warfarin
Describe the possible novel roles for vitamin K beyond its role in coagulation.
While nutrients such as vitamin E, vitamin C and carotenoids have received substantial attention in the media and the medical literature, vitamin K is seldom mentioned. Traditionally thought to be important only for its role in coagulation, recent advances in vitamin K research suggest that it may have an important role in bone health and vascular disease. The new research also suggests a potentially important interaction between vitamins K and E and, further, that food processing may result in alterations in vitamin K form and function.
Vitamin K: What Is It and Where Does It Come From?
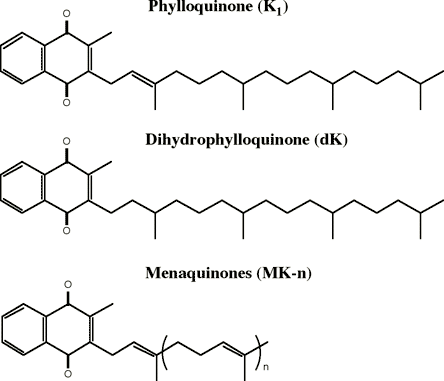
Vitamin K is a fat soluble vitamin found in a variety of food sources. Vitamin K refers to a family of compounds (Figure 1), the most common consumed form being phylloquinone (vitamin K1) which is found in plants.
Figure 1. Forms of Vitamin K.
Green leafy vegetables are among the richest sources; vegetables account for approximately 60% of vitamin K intake in the US. Vitamin K is also found in some plant oils and products derived from plant oils such as salad dressing or margarine, as well as in an increasing number of dietary supplements, though in low amounts.
When phylloquinone-containing vegetable oils such as soybean, canola or cottonseed are hydrogenated during food processing, a hydrogenated form of vitamin K, dihydro-vitamin K, is also formed. High concentrations of dihydro-vitamin K occur in processed foods, especially fast food French fries, doughnuts and potato chips. As discussed below, this dihydro-vitamin K (dK) form may have less biologic activity in some tissues compared to its parent compounds.
Non-plant forms of vitamin K, the menaquinones (MK), are present in some animal foods and in products derived from bacterial fermentation [e.g., milk, meats, certain organs (liver), fermented soybean products and fermented cheeses]. Menaquinones share the same active ring structures as phylloquinone but differ in the length and degree of saturation of the repeating isoprenoid units in the side chain. The menaquinones are classified based on the number of repeating units they contain. For example, MK-4 has four repeating units.
It has long been believed that approximately half of human vitamin K is derived from our gut flora, but more recent evidence suggests that most of our vitamin K is derived exogenously and that bacterial sources within the gut are far less important than previously believed. In addition, in certain tissues, MK-4 is may be converted from vitamin K1 obtained from the diet.
What Are the Biologic Actions of Vitamin K?
As previously mentioned, vitamin K has long been recognized as necessary for coagulation. Vitamin K acts as a cofactor in the post-translational carboxylation of certain glutamic acid (Glu) residues in approximately 12 proteins (Figure 2). The resulting amino acid formed, γ-carboxyglutamic acid (Gla), in the presence of vitamin K has calcium-binding properties that confer function to all vitamin K-dependent proteins, also known as Gla-containing proteins.
Figure 2. The Vitamin K Cycle.
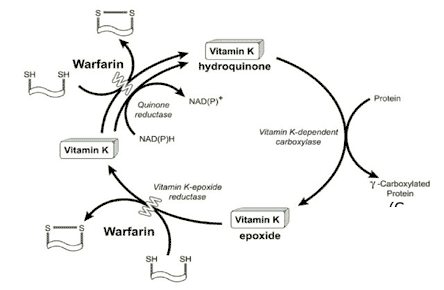
Several vitamin K-dependent hepatic proteins have roles in coagulation (factors II, VII, IX, X, Protein C, Protein S). In the absence of vitamin K, or when the actions of vitamin K are antagonized by medications such as warfarin, these proteins have incomplete carboxylation of the Gla residues, resulting in impaired function. As depicted in Figure 2, the normal vitamin K cycle includes reduction of dietary vitamin K to the biologically active hydroquinone, which then acts as a cofactor to the γ-carboxylation reaction of these vitamin K-dependent proteins. The resulting vitamin K epoxide is then reduced to a quinone that is reused in the γ-carboxylation reaction. Warfarin binds to the epoxide reductase, which interferes with normal recycling of the vitamin.
Several extrahepatic proteins also require vitamin K for their function, as mediated by the formation of Gla residues. Several of these proteins are found in bone, including osteocalcin (OC), matrix Gla protein (MGP) and Protein S. Osteocalcin is a protein produced in osteoblasts and has been used as an indicator of bone formation. The degree to which osteocalcin is carboxylated is sensitive to vitamin K nutriture, and the ratio of undercarboxylated to carboxylated osteocalcin correlates well with vitamin K intake. Osteocalcin, which is the most abundant noncollagenous protein in bone, binds to the hydroxyapatite in bone, suggesting that vitamin K may have a role in promoting bone mineralization.
Matrix Gla Protein is also present in other tissues such as cartilage and vascular walls. There is some indication that MGP may have a role in the regulation of calcification of these tissues. Other tissues with relatively high levels of vitamin K and vitamin K-dependent proteins include the central nervous system (CNS). Most of the research that pertains to the potential role of vitamin K in the CNS is limited to in vitro and animal models, with limited data in humans.
How Is Vitamin K Status Measured?
The classic measure of vitamin K deficiency involves prothrombin (factor II), a vitamin K-dependent protein. However, more recent research indicates that the measurement of prothombin time (PT) is in fact an insensitive measure of vitamin K deficiency. It is estimated that at least 60% of the prothrombin molecules need to be uncarboxylated (i.e., not carboxylated from Glu to Gla) before the PT is prolonged. So while a PT is appropriate for monitoring the anticoagulated patient, more sensitive measures of undercarboxylation such as PIVKA-II (Proteins Induced by Vitamin K Absence or Antagonism - Factor II) are appropriate for individuals without frank deficiency. Likewise, one can measure the vitamin K epoxide that accumulates secondary to warfarin antagonism of the vitamin K cycle but, similar to PIVKA-II, it is only available as a research assay at this time. For measurement of vitamin K status in extra-hepatic tissues such as bones, serum % undercarboxylated osteocalcin appears to be the most sensitive of indicators currently available.
When serum or plasma vitamin K are measured, the level generally reflects recent intake -- depletion or supplementation can lead to marked changes over a period of just a few days. There are also non-dietary factors that influence circulating levels of vitamin K, the most significant being the plasma level of triglycerides. Because vitamin K is primarily transported on the triglyceride-rich chylomicrons, circulating vitamin K levels tend to be elevated in the hyperlipidemic patient. For reasons not clearly understood, between 10-15% of the general adult population have low or undetectable vitamin K levels, despite having normal body stores and normal coagulation function. Therefore, a single phylloquinone determination is often an unreliable indicator of vitamin K status. Body stores of vitamin K also appear to be lower than those for other fat-soluble vitamins, presumably because vitamin K is continually being recycled. Further, each tissue appears to preferentially store different forms or metabolites (Table 1). This variable distribution may provide clues as to the role of vitamin K in that tissue, as will become apparent below.
Table 1. Vitamin K Forms in Tissues.
| In Plasma | Phylloquinone (predominant form) Menaquinones (MK-4, MK-7, MK-8) |
| In Bone | Phylloquinone (predominant form) Menaquinones (MK-4 through MK-8) |
| In Liver | Menaquinones (MK-7 though MK-13) Phylloquinone (minor form) |
Is Vitamin K Good for Bones?
Observational studies consistently demonstrate a relationship between vitamin K and bone health in older people.(1) In these studies, both habitual vitamin K intake as well as biologic markers of vitamin K sufficiency have been examined in relation to bone mineral density (BMD) and fracture risk. In the Framingham Offspring Study, women in the top quartile of vitamin K intake had significantly greater BMD than those with lower intake.(2) Such observations are consistent with in vitro and animal studies, suggesting a role for vitamin K in the promotion of bone mineral deposition.
There are few studies published to date investigating the influence of vitamin K supplementation on bone. Braam et al. compared supplementation of vitamin K1 (1 mg/day), vitamin D and calcium with supplementation consisting only of vitamin D and calcium in postmenopausal women.(3) The group receiving vitamin K with calcium and vitamin D had reduced bone loss at the hip (but not spine) over three years. In another trial, supplementation with pharmacological doses of MK-4 (45 mg/day) resulted in increased lumbar BMD and reduced fracture rate over 24 month.(4) However, it should be noted that the latter trial used pharmacological doses of MK-4, which is not consumed in high amounts in the diet.
Indeed the recommended intakes for vitamin K are currently set at 90 - 120 μg/d, and in vitro evidence does suggest that pharmacological doses of MK-4 have a direct effect on bone resorption attributable to mechanisms other than vitamin K- dependent γ-carboxylation. While these first studies are promising, more clinical trials using physiological doses of vitamin K are required before conclusions can be drawn regarding the role of vitamin K on bone. It should also be emphasized that these studies need to compare the effectiveness of vitamin K supplementation in reduction of age-related bone loss in individuals consuming adequate vitamin D and calcium intakes.
A Role for Vitamin K in Coronary Heart Disease and Vascular Calcification?
In several epidemiologic studies, an inverse relationship between vitamin K intake (or indicators of vitamin K status) and coronary heart disease (CHD) was noted.(3),(5),(6) While this relationship appears to be independent of several other dietary or lifestyle factors associated with CHD, it remains unclear if a true causal relationship exists. For example, green leafy vegetables are the primary sources of dietary vitamin K in the US and Europe, thus vitamin K status may reflect healthy diets or lifestyles more than a specific factor associated with CHD risk.(6)
One potential mechanistic link between vitamin K and atherosclerosis is arterial calcification. In vitro and in vivo evidence suggest that interruption of vitamin K biological function results in abnormal arterial calcification. Specifically, interference with MGP function induced by vitamin K deficiency appears to be an important factor in calcification. In mice, knockout of the MGP gene results in 50% fetal death, and in live births there is marked calcification of the arterial tree as well as osteopenia. Human data are limited primarily to case reports of individuals exposed to warfarin in utero. However, one recent study in postmenopausal women suggested that carotid artery calcification is inversely related to indicators of vitamin K status. This is an area of research that is anticipated to expand in the near future.
Does Vitamin E Interfere with Vitamin K?
Vitamin E has previously been reported to potentiate the effect of warfarin on anticoagulation. The mechanism is unknown, although it has been suggested that vitamin E may impair absorption of vitamin K, or that tocopherol quinone, a vitamin E metabolite, competes with the vitamin K hydroquinone, thereby further reducing the vitamin K hydroquinone's capacity to act as a cofactor for the γ-carboxylase in the anticoagulated patient. However, in vitamin E supplementation trials in individuals with normal coagulation status, there have been no consistent reports of hemorrhagic complications, and trials specifically assessing the influence of vitamin E supplementation on coagulation have not noted a consistent effect.
Two 2004-reported studies in which vitamin E 1000 IU was provided daily for 12 weeks to adults with normal coagulation may help resolve this confusion.(7) After 12 weeks of supplementation, PIVKA-II measures significantly increased in both studies, which included men and women of different ages. In contrast, plasma phylloquinone and undercarboxylated OC were not affected by vitamin E supplementation in either study; though neither study controlled for dietary intakes of vitamin K, which influence both these parameters. In the one study in which PT was measured, there were no changes in PT nor were there adverse bleeding events in the supplemented group.
These results suggest that vitamin E does not interfere with vitamin K absorption but that there is a specific inhibition of hepatic vitamin K-dependent proteins or, more specifically, of prothrombin. Because this is the first study to demonstrate an interference of vitamin K by vitamin E supplementation in adults of normal coagulation, more research is required to elucidate the mechanism.
Conclusions
Current research suggests that vitamin K may have a protective role in bone and vascular health. In the latest US recommendations, the Adequate Intake (AI) recommendation of 120 mcg/d for men and 90 mcg/d for women was based on the relationship between dietary vitamin K and maintenance of normal coagulation proteins. The optimal intake needed for bone health, or prevention of atherosclerotic disease, remains undefined. For persons not taking warfarin, a diet rich in vitamin K-containing foods is recommended, which is likely to be high in other naturally occurring vitamins, minerals and antioxidants.
To date, there is no evidence that high intake of vitamin K from food has deleterious consequences, including promoting thrombosis. For patients taking warfarin, elimination of vitamin K from the diet may facilitate stabilization of anticoagulation but may also have long-term deleterious consequences. An alternative recommendation would be to encourage daily consumption of a consistent level of vitamin K intake. To assist practitioners and patients, values of the vitamin K content of foods are continuously updated and are available (www.ars.usda.gov/ba/bhnrc/ndl). Practitioners and patients should also carefully read dietary supplement labels, as an increasing number of supplements now contain vitamin K.